The landscape of modern psychiatry has been fundamentally altered by the emergence of ketamine as a rapid-acting intervention for treatment-resistant conditions. However, as clinical adoption accelerates, concerns regarding its long-term physiological safety have intensified. A comprehensive systematic review recently published in the Journal of Psychopharmacology provides a nuanced assessment of these risks, specifically focusing on urological and renal complications. The findings suggest that while medically supervised, subanesthetic doses of ketamine do not appear to pose an immediate threat of severe bladder damage, the medical community remains cautious due to significant gaps in long-term data and the limitations of current monitoring protocols.
The Evolution of Ketamine: From Anesthetic to Antidepressant
Ketamine’s journey into the medical mainstream began in 1962 when it was synthesized by organic chemist Calvin Stevens at Parke-Davis as a safer alternative to phencyclidine (PCP). Approved by the U.S. Food and Drug Administration (FDA) in 1970, it quickly became a staple anesthetic on the battlefields of the Vietnam War due to its unique "dissociative" properties, which allowed for profound analgesia without suppressing respiratory or cardiovascular function.
For decades, ketamine was categorized almost exclusively as an anesthetic and a drug of abuse. However, the turn of the millennium marked a pivotal shift. In 2000, researchers at Yale University published a landmark study demonstrating that low-dose intravenous ketamine could alleviate symptoms of major depressive disorder within hours, rather than the weeks required for traditional selective serotonin reuptake inhibitors (SSRIs). This discovery paved the way for the 2019 FDA approval of esketamine (Spravato), a potent nasal spray derivative, and the subsequent explosion of "off-label" ketamine clinics across the globe.
Today, the drug is utilized to treat a spectrum of conditions, including Treatment-Resistant Depression (TRD), Post-Traumatic Stress Disorder (PTSD), and generalized anxiety. Unlike traditional antidepressants that target the monoamine system (serotonin and norepinephrine), ketamine acts primarily as an N-methyl-D-aspartate (NMDA) receptor antagonist. By blocking these receptors, it triggers a cascade of neurobiological events that enhance synaptic plasticity and "rewire" neural circuits associated with mood regulation.
Understanding Ketamine-Induced Urological Toxicity
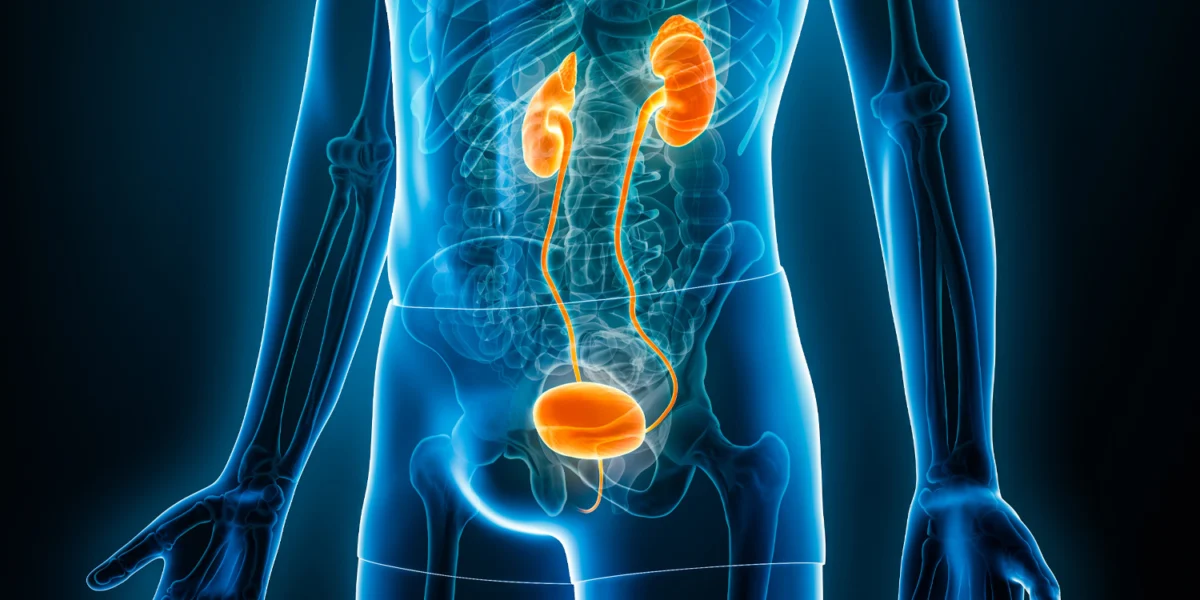
The primary impetus for the systematic review conducted by Jess Kerr-Gaffney and her team at King’s College London was the well-documented phenomenon of "ketamine-induced urological toxicity" observed in recreational users. In the context of chronic, high-dose illicit use, ketamine metabolites are excreted through the urinary tract, where they can cause direct toxic effects on the bladder’s epithelial lining (the urothelium).
Clinical observations of recreational users have revealed a harrowing progression of symptoms:
- Bladder Wall Thickening: Chronic inflammation leads to scarring and the contraction of the bladder wall.
- Reduced Capacity: The organ loses its ability to expand, leading to a constant and painful urge to urinate.
- Hematuria: Damage to the lining often results in blood in the urine.
- Renal Failure: In extreme cases, the back-pressure of urine and the spread of inflammation can cause hydronephrosis and permanent kidney damage.
While psychiatric doses are significantly lower than those used by recreational "heavy users"—who may consume several grams a day—the medical community has worried that the repeated, long-term maintenance dosing required for psychiatric stability could produce a cumulative effect similar to recreational toxicity.
Methodology and Findings of the Systematic Review
To address these concerns, the researchers at King’s College London performed a rigorous search of medical databases, filtering thousands of papers to identify 27 studies that specifically tracked urological outcomes in adult psychiatric patients. The cohort primarily consisted of individuals with severe depressive disorders, though some studies included patients with anxiety and trauma-related conditions.
The results of the review indicate a high degree of variability in reported side effects. Across the 27 studies, the incidence of urinary symptoms ranged from 0% to approximately 25%. When patients did report issues, they were generally classified as mild or moderate. The most frequent complaints included:
- Increased urinary frequency: The need to urinate more often than usual.
- Dysuria: A burning sensation or discomfort during urination.
- Urinary urgency: A sudden, compelling need to void the bladder.
- Polyuria: The production of abnormally large volumes of urine.
Crucially, the review found that in randomized controlled trials (RCTs), the rate of urological complaints among those receiving ketamine was often indistinguishable from those in the control groups receiving a placebo. This suggests that some reported symptoms may be related to the underlying psychiatric condition, concurrent medications, or the "nocebo effect," where a patient’s expectation of side effects leads to their perception.
The Esketamine Distinction and Objective Testing
The review highlighted specific data regarding esketamine, the purified S-enantiomer of ketamine. In three separate trials involving the nasal spray, patients reported slightly higher rates of urinary symptoms compared to placebo groups. While these results did not reach the threshold of statistical significance, they suggest that the higher potency or specific delivery method of esketamine may warrant closer urological scrutiny.
Furthermore, several studies included in the review utilized objective laboratory markers rather than relying solely on patient self-reporting. By analyzing urine for red blood cells (microscopic hematuria), white blood cells (indicating inflammation or infection), and protein levels, researchers were able to look for "silent" damage. These laboratory measurements remained remarkably stable across the treatment durations studied, providing an extra layer of reassurance that cellular damage was not occurring undetected in the short term.
Critical Limitations: The "Time-Lag" Problem
Despite the generally positive findings, the authors of the review issued a stern warning regarding the limitations of the current evidence base. The most significant concern is the timeframe of clinical monitoring. The median follow-up period across the 27 studies was only four weeks.
Urological toxicity is typically a chronic condition that develops over months or years of exposure. A one-month snapshot is insufficient to determine the safety of maintenance therapy, which many psychiatric patients utilize for years to prevent relapse. Only five of the reviewed studies followed patients for longer than six months, leaving a massive data gap regarding the long-term cumulative effects of subanesthetic ketamine on the human bladder.
The Risks of Passive Monitoring and the "Analgesic Mask"
The review also exposed systemic flaws in how clinical trials monitor physical safety. Eleven of the studies relied on "passive monitoring," a method where clinicians only record side effects if the patient proactively volunteers the information. This approach is notoriously unreliable for several reasons:
- Patient Dismissal: Patients may attribute mild bladder irritation to other factors, such as aging, diet, or caffeine intake.
- Stigma: Patients may be reluctant to discuss urinary or "private" health issues with a psychiatric provider.
- The Analgesic Mask: Ketamine is a powerful painkiller. If the drug is actively numbing the patient’s pelvic region, they may not feel the early, subtle pain of bladder inflammation. By the time the medication wears off and the pain becomes noticeable, the tissue damage may already be advanced.
Furthermore, the review noted a high risk of bias in many trials. Many were "unblinded," meaning both the patient and the doctor knew the active drug was being administered. This lack of blinding can skew the reporting of both benefits and side effects.
Implications for the Rise of At-Home Ketamine
The findings of the King’s College London team arrive at a critical juncture for the healthcare industry. The COVID-19 pandemic accelerated the rise of telehealth, leading to the emergence of medical startups that prescribe ketamine for at-home use. In these models, patients often receive compounded ketamine lozenges (troches) or rapid-dissolve tablets via mail.
Unlike in-clinic IV infusions or esketamine nasal spray administered under direct supervision, at-home use often lacks rigorous physical monitoring. Patients may be taking the drug more frequently or for longer durations without ever undergoing a urinalysis or a standardized urological screening. This "Wild West" of psychiatric treatment creates a scenario where early signs of organ damage could be missed entirely, leading to a potential public health crisis of ketamine-induced cystitis in the coming decade.
Recommendations for Future Clinical Practice
To mitigate these risks, the research team recommended a fundamental shift in how ketamine treatment is managed. They proposed that all future clinical trials and active clinical practices adopt the following protocols:
- Mandatory Standardized Screening: Instead of passive monitoring, clinicians should use validated tools, such as the Pelvic Pain and Urgency/Frequency (PUF) Scale or the O’Leary-Sant Interstitial Cystitis Symptom Index, on a weekly or bi-weekly basis.
- Baseline and Routine Urinalysis: Patients should undergo a simple, non-invasive urine test before beginning treatment and at regular intervals (e.g., every three months) during maintenance therapy to check for microscopic biomarkers of inflammation.
- Interdisciplinary Collaboration: There should be a clearer pathway for psychiatric clinics to refer patients to urologists at the first sign of persistent urinary changes.
Conclusion: A Balanced Perspective
The systematic review authored by Jess Kerr-Gaffney and colleagues serves as both a reassurance and a cautionary tale. For patients currently benefiting from ketamine for life-threatening depression, the data suggests that the risk of severe bladder damage from controlled, short-term medical use is low. However, the "absence of evidence is not evidence of absence."
As ketamine moves from a niche experimental treatment to a mainstream psychiatric tool, the medical community must move beyond the "honeymoon phase" of its antidepressant success and commit to long-term physiological vigilance. Protecting the physical health of psychiatric patients is as paramount as treating their mental distress, and only through rigorous, long-term monitoring can the true safety profile of this powerful chemical be fully understood.