Researchers at the University of Illinois Urbana-Champaign have unveiled a groundbreaking method for charting the intricate web of connections between brain cells, a development poised to revolutionize neuroscience. The innovative technique, dubbed Connectome-seq, utilizes molecular "barcodes" to tag individual neurons, allowing scientists to map neural connections with remarkable speed and unprecedented single-synapse resolution. This advancement promises to significantly deepen our understanding of how complex brain networks are organized, how they function, and what goes awry in devastating neurological disorders.
The challenge of mapping the brain’s circuitry has long been a formidable obstacle for scientists. Traditional methods involved laborious processes of slicing brain tissue into ultrathin sections, imaging these slices under powerful microscopes, and then painstakingly piecing together the neural pathways, a process akin to assembling an infinitely complex jigsaw puzzle. While more recent sequencing-based tools have enabled the simultaneous labeling of numerous neurons, they have historically fallen short of identifying the precise synaptic connections between them.
"When engineering a computer, you need to know the circuitry of the central processing unit. If you don’t know how everything is wired together, you can’t understand its function, optimize it or fix it when something breaks. We are approaching the brain the same way," explained study leader Boxuan Zhao, a professor of cell and developmental biology at the University of Illinois Urbana-Champaign. Zhao’s team aimed to bridge this critical gap, developing a technology that could provide a comprehensive and detailed blueprint of neural communication.
The Genesis of Connectome-seq: A Novel Approach
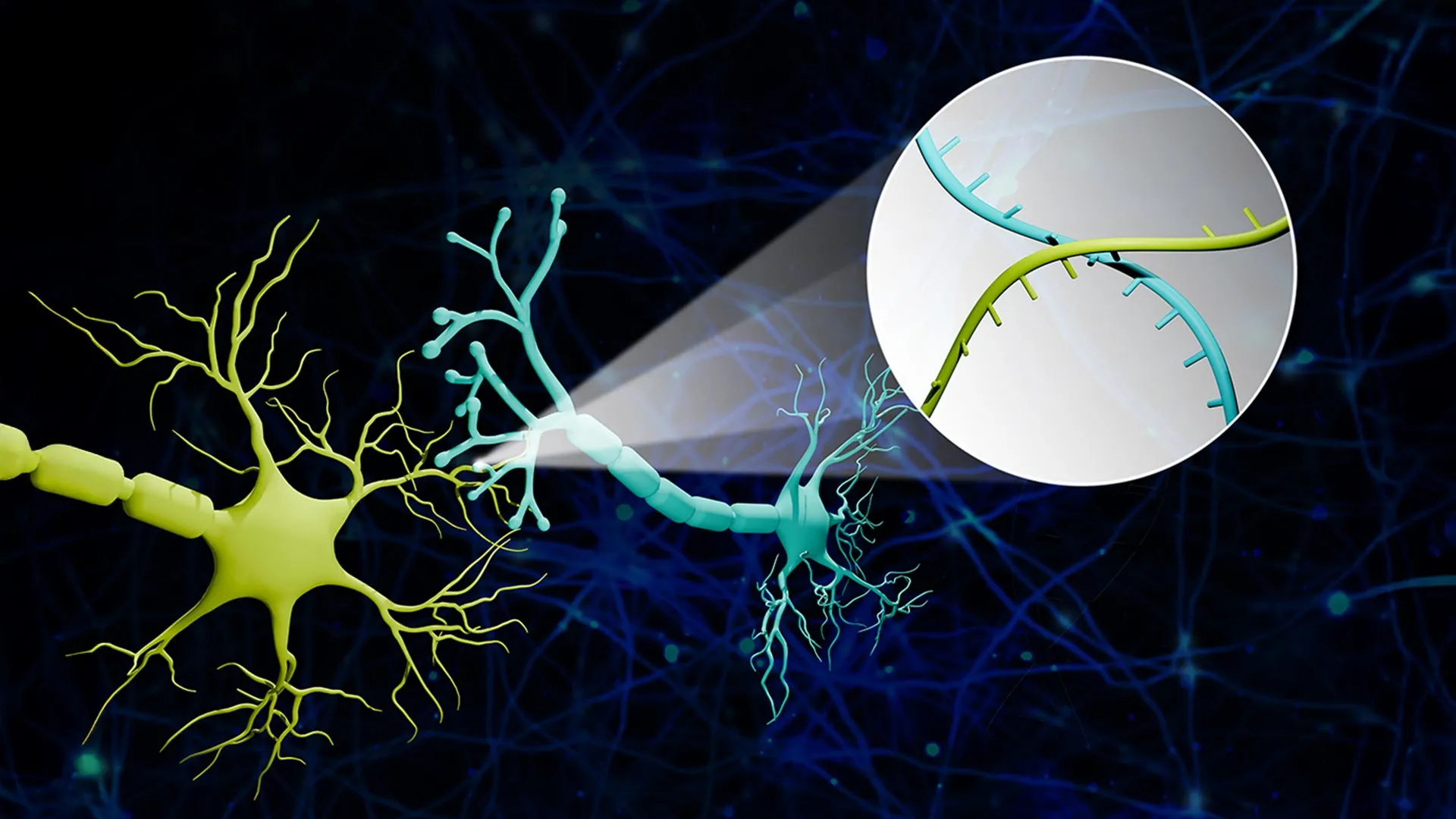
The development of Connectome-seq represents a paradigm shift in neural mapping. The core of this technology lies in its ability to assign each neuron a unique RNA "barcode." These barcodes are not merely passive markers; they are actively transported by specialized proteins from the neuron’s soma (its main body) to the synapse, the crucial junction where one neuron communicates with another.
Once these tagged synapses are isolated, the researchers employ high-throughput sequencing to read the barcode pairs present. The presence of specific barcode pairs together at a synapse directly reveals which two neurons are making a direct connection. This innovative approach transforms the complex biological problem of neural connectivity into a tractable sequencing challenge, enabling scientists to map neural networks on a vast scale.
Zhao further elaborated on the analogy, stating, "We translated the neural connectivity problem into a sequencing problem. Imagine a big bunch of balloons. The main body of each balloon has its unique barcode stickers all over it, and some move down to the end of the string. If two balloons are tied together at the end, the two barcodes meet at the junction. Then we snip out the knots and sequence the barcodes in each one. If the same knot has stickers from balloon A and balloon B, we know these two balloons are tied together. We are doing this in the brain, just on the level of thousands of neuron cells. With this information, we can reconstruct a sophisticated map that represents the connections among all these seemingly floaty groups."
Early Successes and Unveiling New Connections
The initial application of Connectome-seq has already yielded significant insights. In their study, published in the prestigious journal Nature Methods, Zhao’s team successfully mapped the connections of over 1,000 neurons within the pontocerebellar circuit of a mouse brain. This specific circuit plays a vital role in coordinating motor control and sensory information between the cerebellum and the pons.
The detailed mapping revealed previously unknown patterns of connectivity within this circuit. Notably, the researchers identified direct links between cell types that were not previously understood to be directly connected in the adult brain. These unexpected connections suggest a more intricate and dynamic organization of neural circuits than previously hypothesized, underscoring the power of Connectome-seq to uncover hidden architectural features of the brain.
"Our technology enables simultaneous mapping of thousands of neural connections with single-synapse resolution — a capability that doesn’t exist in any current technology," Zhao emphasized. "It is directly applicable to understanding circuit dysfunction in neurodegenerative diseases and could provide a platform for developing circuit-guided therapeutic interventions."
A Timeline of Advancement in Brain Mapping
The quest to map the brain has been a long and arduous journey, marked by incremental advancements:
- Early Microscopy (Late 19th – Early 20th Century): Pioneering neuroanatomists like Santiago Ramón y Cajal used staining techniques to visualize individual neurons and their general dendritic and axonal projections, laying the foundation for understanding neuronal structure. However, detailed synaptic connections remained largely elusive.
- Electron Microscopy (Mid-20th Century onwards): The advent of electron microscopy allowed for visualization of synapses at a much higher resolution, enabling the identification of synaptic structures. However, this technique was still limited to small tissue volumes and was not amenable to large-scale circuit mapping.
- Genetic and Molecular Tracing Techniques (Late 20th – Early 21st Century): Advances in genetic engineering and molecular biology led to tools like viral tracers and fluorescent proteins, which could label and track neuronal pathways. While powerful, these methods often provided information about axonal projections rather than precise synaptic partners.
- High-Throughput Sequencing Approaches (21st Century): The development of next-generation sequencing technologies opened doors for analyzing large amounts of genetic material. Researchers began to adapt these tools for tracing neural connections, but often with limitations in spatial resolution or the ability to pinpoint individual synapses.
- Connectome-seq (Present): The Connectome-seq platform represents the latest leap forward, integrating molecular barcoding with high-throughput sequencing to achieve single-synapse resolution across thousands of neurons simultaneously.
Zhao’s team is already looking towards the future, with ongoing improvements to the Connectome-seq platform. "With improvements already underway in our lab, we are confident that we can make it even better and eventually reach the goal of mapping the whole mouse brain," Zhao stated, signaling a future where comprehensive brain atlases become a reality.
Implications for Neurological Disorders and Therapeutics
The potential impact of Connectome-seq extends far beyond fundamental neuroscience research. Its speed, scalability, and unparalleled detail make it a powerful tool for understanding and combating a wide range of neurological and psychiatric conditions.
By enabling researchers to precisely map neural circuits, Connectome-seq offers a novel approach to dissecting the complexities of neurodegenerative diseases such as Alzheimer’s, Parkinson’s, and Huntington’s disease. Scientists can now compare the connectivity patterns in healthy brains with those affected by disease at various stages, potentially identifying subtle yet critical alterations in neural networks that precede the onset of overt symptoms.
"With sequencing-based approaches, the time and cost are greatly reduced, which really makes it possible to see differences in different brains. We could see where connections change, where the most vulnerable parts of the brain are, perhaps before symptoms even appear," Zhao explained.
This detailed understanding of circuit dysfunction could pave the way for the development of highly targeted therapeutic interventions. For instance, in the context of Alzheimer’s disease, researchers might be able to pinpoint the exact weak link in a neural circuit that initiates the cascade of neurodegeneration. Armed with this knowledge, they could explore strategies to specifically strengthen these vulnerable connections, potentially slowing or even halting disease progression.
Furthermore, Connectome-seq could illuminate the underlying circuit abnormalities in psychiatric disorders like schizophrenia, depression, and anxiety. By providing a detailed map of how brain regions communicate, it may become possible to identify specific dysfunctions that contribute to these complex conditions, leading to more effective and personalized treatments.
Broader Impact and Future Directions
The development of Connectome-seq is a testament to the power of interdisciplinary research, combining expertise in molecular biology, genomics, and computational neuroscience. The successful application in mice suggests that the technology can be adapted for use in other model organisms, and eventually, for studying the human brain.
The implications for drug discovery are also significant. By understanding how drugs affect neural circuits at a granular level, researchers can develop more effective and less toxic therapeutics. This could accelerate the pipeline for new treatments for a multitude of brain disorders.
The research was made possible through substantial funding from various institutions, including a Neuro-omics Initiative grant from the Wu Tsai Neurosciences Institute of Stanford University, and support from the Elsa U. Pardee Foundation and the Edward Mallinckrodt Jr. Foundation. This collaborative effort underscores the growing recognition of the importance of understanding the brain’s complex wiring.
While the complete mapping of the entire human brain remains a distant goal, Connectome-seq represents a monumental stride in that direction. The ability to rapidly and accurately chart neural connections at the single-synapse level opens up unprecedented avenues for scientific inquiry and therapeutic innovation, offering a renewed sense of hope for millions affected by neurological and psychiatric conditions worldwide. The era of detailed brain circuit mapping has truly begun.