A groundbreaking advancement in neuroscience is poised to revolutionize our understanding of the brain’s intricate wiring. Researchers at the University of Illinois Urbana-Champaign have unveiled a novel method that utilizes molecular "barcodes" to tag individual neurons, enabling the rapid and highly detailed mapping of neural connections. This innovative technique has already allowed scientists to chart thousands of connections within the mouse brain with a level of precision previously unattainable, opening new avenues for deciphering brain function, diagnosing neurological disorders, and developing targeted therapies.
The Challenge of Mapping the Brain’s Complex Architecture
For decades, neuroscientists have grappled with the monumental task of mapping the brain’s connectome – the complete set of neural connections. This endeavor is akin to understanding a city’s infrastructure by examining every road, intersection, and traffic flow simultaneously. Traditional methods have been labor-intensive and time-consuming, often involving the meticulous slicing of brain tissue into infinitesimally thin sections, followed by laborious microscopic imaging and manual reconstruction of neural pathways. While advancements in sequencing-based tools have offered the ability to label multiple neurons at once, they typically indicate where a neuron extends, rather than precisely identifying the specific synapses – the critical junctions where communication occurs between neurons.
"When engineering a computer, you need to know the circuitry of the central processing unit," explained Boxuan Zhao, a professor of cell and developmental biology at the University of Illinois Urbana-Champaign and the study’s lead investigator. "If you don’t know how everything is wired together, you can’t understand its function, optimize it, or fix it when something breaks. We are approaching the brain the same way." This analogy highlights the fundamental need for a comprehensive wiring diagram to unlock the brain’s mysteries.
Introducing Connectome-seq: A Paradigm Shift in Neural Mapping
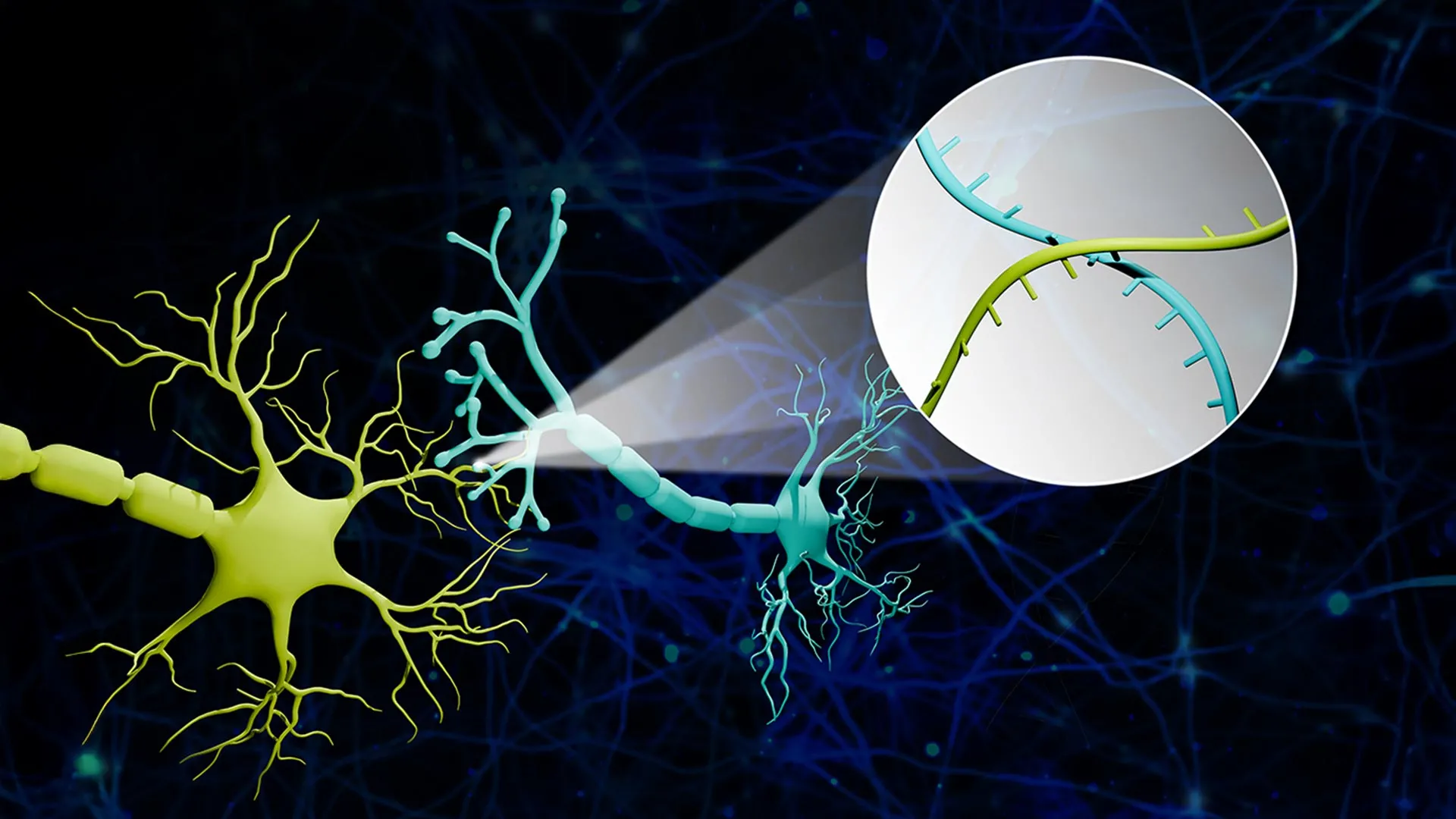
To surmount these limitations, Zhao’s team engineered a sophisticated platform they named Connectome-seq. The core innovation lies in assigning each neuron a unique RNA "barcode." These molecular identifiers are then strategically transported by specialized proteins from the neuron’s cell body to its axon terminals, reaching the synapse – the microscopic gap where electrochemical signals are transmitted between neurons.
The process then shifts to the realm of advanced molecular biology and high-throughput sequencing. Researchers meticulously isolate these synapses. By employing sophisticated sequencing techniques, they can then read the barcode pairs present at each junction. The co-occurrence of specific barcode pairs directly indicates which neurons are forming direct connections. This elegant solution translates the complex biological problem of neural connectivity into a tractable sequencing challenge, allowing for the mapping of neural networks on an unprecedented scale.
Zhao further elaborated on the ingenuity of the method using a vivid analogy: "We translated the neural connectivity problem into a sequencing problem. Imagine a big bunch of balloons. The main body of each balloon has its unique barcode stickers all over it, and some move down to the end of the string. If two balloons are tied together at the end, the two barcodes meet at the junction. Then we snip out the knots and sequence the barcodes in each one. If the same knot has stickers from balloon A and balloon B, we know these two balloons are tied together. We are doing this in the brain, just on the level of thousands of neuron cells. With this information, we can reconstruct a sophisticated map that represents the connections among all these seemingly floaty groups."
Unveiling Novel Neural Circuitry in the Mouse Brain
The initial application of Connectome-seq in a mouse brain circuit, specifically the pontocerebellar circuit that bridges two major brain regions, yielded remarkable results. The analysis successfully mapped over 1,000 neurons, uncovering previously undocumented patterns of connectivity. Crucially, the technique revealed direct links between cell types that were not previously known to interact in the adult brain. These discoveries underscore the power of Connectome-seq to reveal hidden organizational principles within neural circuits.
The implications of such detailed mapping are profound. Understanding these intricate connections is fundamental to deciphering how the brain processes information, generates thoughts, and controls behavior. Deviations from these established pathways are often implicated in a wide range of neurological and psychiatric conditions.
Accelerating Research into Neurodegenerative Diseases and Beyond
The speed and scalability of Connectome-seq hold immense promise for accelerating research into a host of debilitating brain disorders, including Alzheimer’s disease, Parkinson’s disease, schizophrenia, and depression. By enabling researchers to rapidly compare the connectomes of healthy brains with those affected by disease at various stages, scientists can identify subtle yet critical changes in neural circuits that may precede the onset of clinical symptoms.
"Our technology enables simultaneous mapping of thousands of neural connections with single-synapse resolution – a capability that doesn’t exist in any current technology," stated Zhao. "It is directly applicable to understanding circuit dysfunction in neurodegenerative diseases and could provide a platform for developing circuit-guided therapeutic interventions."
The potential to identify the earliest molecular markers of disease progression is particularly exciting. "With sequencing-based approaches, the time and cost are greatly reduced, which really makes it possible to see differences in different brains," Zhao explained. "We could see where connections change, where the most vulnerable parts of the brain are, perhaps before symptoms even appear. For example, if we can catch where exactly the weak link is that kick starts the whole catastrophic cascade in Alzheimer’s disease, can we specifically strengthen those connections to where the disease slows or does not progress?" This proactive approach to understanding and treating brain diseases could represent a significant departure from current reactive strategies.
A Look at the Timeline and Future Directions
The development of Connectome-seq is the culmination of years of research in molecular biology, genomics, and neuroscience. While the exact timeline of its conception and development is not detailed in the initial announcement, the publication of these findings in the prestigious journal Nature Methods signifies a significant milestone. The journal Nature Methods is known for publishing rigorous and impactful research that introduces new experimental techniques and analytical approaches. Its selection for publication indicates that the scientific community recognizes the broad applicability and potential of Connectome-seq.
Zhao expressed optimism about the future trajectory of the technology. "With improvements already underway in our lab, we are confident that we can make it even better and eventually reach the goal of mapping the whole mouse brain," he stated. This ambition points towards a future where a complete connectome of the mouse brain, a crucial model organism for human neurological research, could be generated. Such a comprehensive map would serve as an invaluable resource for the entire neuroscience community.
Broader Implications and Expert Perspectives
The development of Connectome-seq has the potential to fundamentally alter how neuroscience research is conducted. By providing a faster, more detailed, and scalable method for mapping neural connections, it democratizes access to powerful analytical tools. This could lead to a surge in discovery across various subfields of neuroscience, from developmental biology to computational neuroscience.
While specific reactions from external parties were not immediately available, the scientific community’s reaction to such a significant methodological advancement is typically one of eager anticipation and critical evaluation. Experts in connectomics and neurotechnology would likely be examining the technical specifications, reproducibility, and potential limitations of Connectome-seq. The ability to resolve connections at the single-synapse level is a particularly coveted achievement.
The implications for drug development are also substantial. Understanding how specific neural circuits are disrupted in diseases could enable the design of more precise and effective therapeutic interventions. Instead of broad-spectrum treatments, future therapies might target specific synaptic connections or neuronal populations, leading to improved efficacy and reduced side effects.
Funding and Support
This pioneering research was supported by significant funding from several esteemed institutions, underscoring its perceived importance. A Neuro-omics Initiative grant from the Wu Tsai Neurosciences Institute of Stanford University provided crucial backing. Additionally, funding from the Elsa U. Pardee Foundation and the Edward Mallinckrodt Jr. Foundation further enabled the researchers to pursue this ambitious project. Such financial support is vital for pushing the boundaries of scientific inquiry and translating innovative ideas into tangible tools.
In conclusion, Connectome-seq represents a significant leap forward in our ability to understand the brain’s intricate wiring. By providing an unprecedentedly fast and detailed method for mapping neural connections, this technology promises to accelerate discoveries in fundamental neuroscience and pave the way for transformative advances in the diagnosis and treatment of neurological disorders. The ongoing refinement and application of Connectome-seq are eagerly awaited by researchers worldwide.