Researchers at Johns Hopkins Medicine, bolstered by a significant new funding award from the National Institutes of Health (NIH), are forging ahead with a groundbreaking study that could redefine our understanding and treatment of Alzheimer’s disease. At the heart of this research lies a humble yet crucial gas produced by a specific protein in the brain, a substance previously known more for its pungent odor than its vital cognitive functions. This multidisciplinary endeavor is meticulously investigating how this gas, hydrogen sulfide, produced by the enzyme Cystathionine γ-lyase (CSE), plays an indispensable role in the intricate mechanisms of memory formation, and crucially, how its deficit may precipitate the devastating cognitive decline associated with Alzheimer’s.
Unveiling the Cognitive Significance of Hydrogen Sulfide
The protein in question, Cystathionine γ-lyase (CSE), is primarily recognized for its role in generating hydrogen sulfide (H₂S), a gas notorious for its "rotten egg" smell. However, this latest research, published in the esteemed journal Proceedings of the National Academy of Sciences, suggests that H₂S, when present at precisely regulated, minute levels within the brain, is far more than a mere olfactory byproduct. It appears to be a critical mediator in the processes that underpin how memories are formed and consolidated. Dr. Bindu Paul, an associate professor of pharmacology, psychiatry, and neuroscience at the Johns Hopkins University School of Medicine and the lead investigator of this pivotal study, explained the genesis of this line of inquiry. "For years, we’ve known that certain gases play signaling roles in the body, but hydrogen sulfide’s association with toxicity often overshadowed its potential physiological benefits. Our work is fundamentally shifting that perception, particularly within the context of brain health."
The research team’s findings are derived from sophisticated experiments conducted on genetically engineered mice, which provide a crucial preclinical model for understanding complex biological processes. These genetically modified rodents were specifically designed to either lack the CSE enzyme or exhibit impaired CSE function, allowing researchers to observe the direct consequences of reduced H₂S production within the brain.
A Timeline of Discovery: From Huntington’s to Alzheimer’s
This latest research is not an isolated event but rather the culmination of years of dedicated investigation by a team at Johns Hopkins Medicine, building upon foundational work laid by luminaries in the field. The intellectual lineage of this project can be traced back to the pioneering efforts of Dr. Solomon Snyder, M.D., D.Sc., D.Phil., a professor emeritus of neuroscience, pharmacology, and psychiatry.
In 2014, Dr. Snyder’s laboratory published seminal findings in Nature that highlighted CSE’s supportive role in brain health, specifically in mouse models of Huntington’s disease, another devastating neurodegenerative disorder. This earlier work utilized mice engineered to be deficient in the CSE protein, a strain first developed in 2008 when the protein’s connection to broader physiological functions, including blood vessel health and blood pressure regulation, was initially elucidated. This established a precedent for exploring CSE’s impact on neurological conditions.
The journey continued in 2021, when the same research group observed a critical dysfunction in CSE within mice exhibiting characteristics of Alzheimer’s disease. More encouragingly, they found that even very small, carefully administered injections of hydrogen sulfide demonstrated a protective effect on brain function in these models. These earlier studies, while highly informative, often focused on mice carrying multiple genetic mutations associated with specific neurodegenerative diseases. The current NIH-funded research distinguishes itself by isolating and meticulously examining the intrinsic role of CSE and H₂S production, independent of other genetic confounding factors.
Dr. Snyder, who retired from the Johns Hopkins Medicine faculty in 2023 but remains a co-corresponding author on the current study, emphasized the significance of this focused approach. "This most recent work indicates that CSE alone is a major player in cognitive function and could provide a new avenue for treatment pathways in Alzheimer’s disease," he stated. "By pinpointing CSE’s independent contribution, we are moving closer to precise therapeutic targets."
The Perilous Link Between Memory Loss and CSE Deficiency
To definitively establish the connection between CSE deficiency and memory impairment, the researchers employed a rigorous experimental design. They meticulously compared mice that completely lacked the CSE protein with their genetically unaltered counterparts, utilizing the same mouse strain that had been instrumental in previous studies. A key component of their investigation involved assessing spatial memory – the ability to remember locations, directions, and environmental cues – using the well-established Barnes maze.
In this particular experimental setup, mice are trained to identify and escape a bright, aversive light by locating a hidden shelter. The results were stark and revealing. At two months of age, both the normal mice and those deficient in CSE performed comparably, successfully locating the shelter within a reasonable timeframe of three minutes. However, a significant divergence emerged by the six-month mark. The CSE-deficient mice began to exhibit considerable difficulty in navigating the maze and finding the escape route, a clear indication of deteriorating spatial memory. In contrast, the normal mice continued to perform at a high level, demonstrating robust spatial memory retention.
"The pronounced decline in spatial memory observed in the CSE-deficient mice strongly suggests a progressive onset of neurodegenerative disease that can be directly attributed to the loss of CSE function," explained Dr. Suwarna Chakraborty, the study’s first author and a researcher in Dr. Paul’s laboratory. This finding provides compelling evidence that the absence of CSE is not merely an incidental observation but a direct contributor to cognitive deficits.
Brain Structure and Function: A Microscopic Mirror of Alzheimer’s
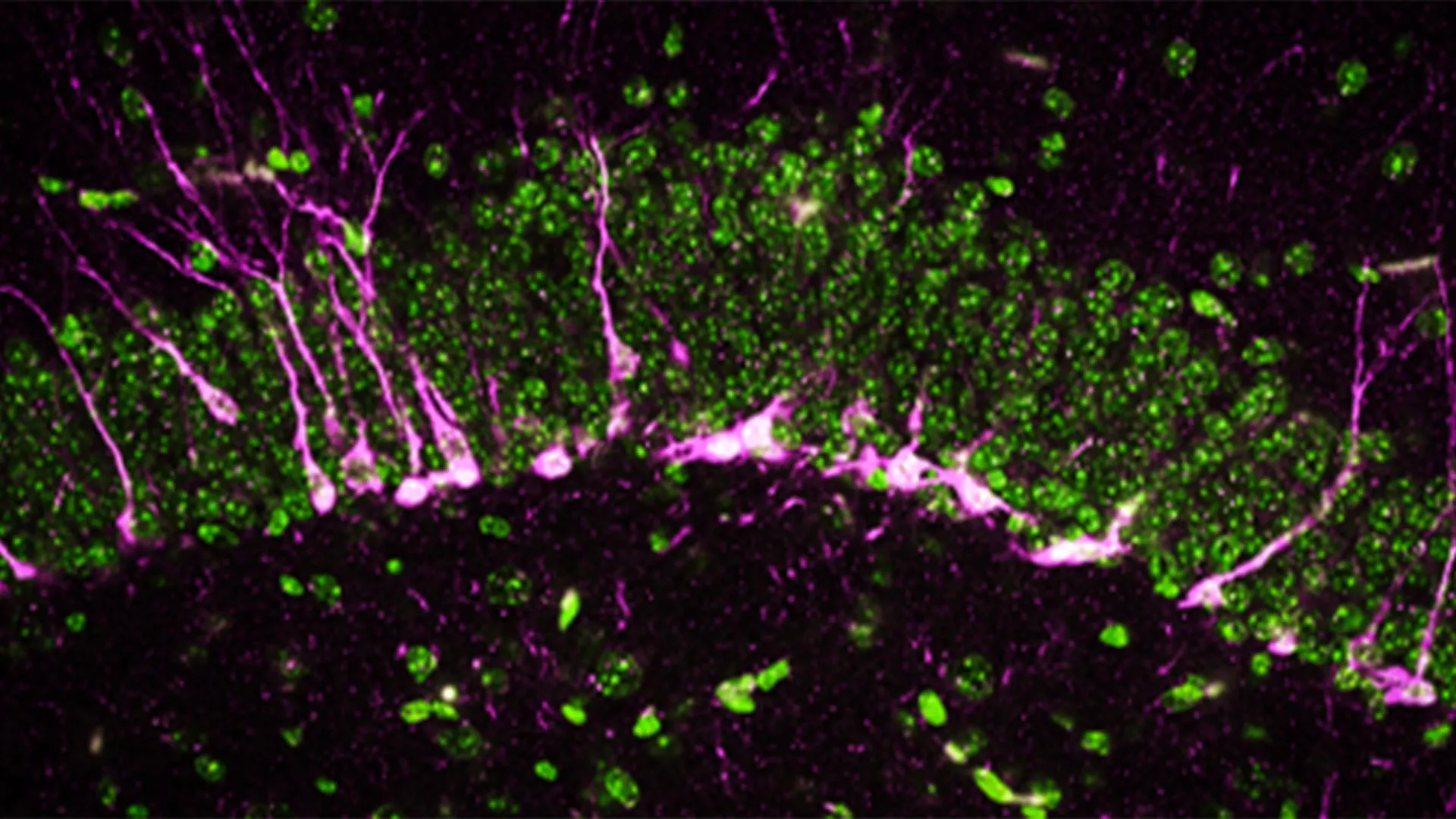
Beyond behavioral assessments, the research team delved into the cellular and structural consequences of CSE absence within the brain. The hippocampus, a brain region critically involved in the formation of new memories and spatial navigation, was a primary focus. A hallmark of healthy brain function, particularly in relation to memory, is the continuous process of neurogenesis – the birth of new neurons. Disruptions in this delicate process are a well-documented characteristic of many neurodegenerative diseases, including Alzheimer’s.
Employing a battery of sophisticated biochemical and analytical techniques, the researchers discovered that key proteins essential for neurogenesis were either significantly reduced in quantity or entirely absent in the brains of mice lacking CSE. This suggests that the fundamental machinery for generating new brain cells was compromised.
Further microscopic examination, utilizing high-powered electron microscopes, revealed tangible structural damage within the brains of these CSE-deficient mice. The scientists observed significant breaks and lesions in the cerebral blood vessels. This damage directly points to a compromised blood-brain barrier (BBB), a vital protective shield that regulates the passage of substances from the bloodstream into the brain. A weakened BBB is another well-established pathological hallmark of Alzheimer’s disease, contributing to neuroinflammation and the accumulation of toxic proteins. Moreover, the study found that newly formed neurons in these mice experienced considerable difficulty in migrating to the hippocampus, the region where they would normally integrate and contribute to memory formation.
"The mice lacking CSE were compromised at multiple levels, which correlated with symptoms that we see in Alzheimer’s disease," stated Dr. Sunil Jamuna Tripathi, a co-first author and researcher in Dr. Paul’s lab. "The confluence of impaired neurogenesis, BBB dysfunction, and difficulties in neuronal migration paints a clear picture of a brain under severe duress, mirroring pathological changes observed in human Alzheimer’s patients."
The Tenuous Balance: Hydrogen Sulfide’s Dual Nature
While the potential of hydrogen sulfide as a neuroprotective agent is exciting, it is imperative to acknowledge its inherent toxicity in higher concentrations. Previous research has consistently demonstrated that while H₂S can protect neurons in mouse models, direct administration of the gas in large quantities poses significant safety risks. This presents a formidable challenge for therapeutic development. The scientific community’s focus has therefore shifted towards understanding how to safely maintain the extremely low, physiological levels of H₂S that are naturally present and beneficial within neurons.
The current research by the Johns Hopkins team is directly addressing this challenge. By investigating the endogenous production of H₂S via CSE, they are exploring ways to bolster the brain’s own capacity to generate this crucial gas, rather than relying on external administration. This approach holds the promise of a more controlled and safer therapeutic strategy.
Broader Implications for Alzheimer’s Treatment
Alzheimer’s disease represents a monumental public health crisis, affecting an estimated 6.7 million Americans aged 65 and older in 2023, according to the Alzheimer’s Association. This number is projected to nearly double by 2050, underscoring the urgent need for effective interventions. Despite decades of research, current treatments primarily focus on managing symptoms and offer limited efficacy in halting or reversing the disease’s relentless progression.
The findings from Johns Hopkins Medicine offer a beacon of hope by identifying a novel therapeutic target. By focusing on CSE and its role in endogenous hydrogen sulfide production, researchers are paving the way for the development of therapies designed to protect brain function and potentially slow or even halt the degenerative cascade characteristic of Alzheimer’s disease. This could involve strategies aimed at enhancing CSE activity, optimizing its molecular environment, or indirectly stimulating H₂S production through other pathways.
A Collaborative Effort Fueled by Diverse Funding
This groundbreaking research was made possible through a robust network of funding sources, highlighting the widespread recognition of its potential impact. Significant support was provided by the National Institutes of Health (NIH) through multiple grants, including 1R01AG071512, P50 DA044123, 1R21AG073684, O1AGs066707, U01 AG073323, AG077396, NS101967, NS133688, and P01CA236778. Additional critical funding came from the Department of Defense (HT94252310443), the American Heart Association, the AHA-Allen Initiative in Brain Health and Cognitive Impairment, the Solve ME/CFS Initiative, the Catalyst Award from Johns Hopkins University, the Valour Foundation, the Wick Foundation, a Department of Veterans Affairs Merit Award (I01BX005976), the Louis Stokes Cleveland Department of Medical Affairs Veterans Center, the Mary Alice Smith Funds for Neuropsychiatry Research, the Lincoln Neurotherapeutics Research Fund, the Gordon and Evie Safran Neuropsychiatry Fund, and the Leonard Krieger Fund of the Cleveland Foundation.
The collaborative spirit of this research is also evident in the diverse group of scientists who contributed to its success. Beyond the lead researchers Dr. Paul and Dr. Snyder, and co-first authors Dr. Chakraborty and Dr. Tripathi, the study involved contributions from Richa Tyagi and Benjamin Orsburn at Johns Hopkins; Edwin Vázquez-Rosa, Kalyani Chaubey, Hisashi Fujioka, Emiko Miller, and Andrew Pieper from Case Western University; Thibaut Vignane and Milos Filipovic from the Leibniz Institute for Analytical Sciences in Germany; Sudarshana Sharma from Hollings Cancer Center; Bobby Thomas from Darby Children’s Research Institute and the Medical University of South Carolina; and Zachary Weil and Randy Nelson from the West Virginia University School of Medicine. This extensive collaboration underscores the complexity of the research and the multifaceted expertise required to tackle such a challenging disease.
The implications of this research are profound. By meticulously unraveling the role of CSE and hydrogen sulfide in memory function and their potential link to Alzheimer’s pathology, the Johns Hopkins team has not only advanced fundamental neuroscience but also illuminated a promising new pathway for therapeutic intervention. As the scientific community continues to build upon these findings, the prospect of developing novel treatments that can effectively combat Alzheimer’s disease moves closer to reality, offering renewed hope to millions worldwide.