A groundbreaking study published in The Lancet Digital Health is shedding new light on the remarkable adaptability of the human brain following a stroke. Researchers at the USC Mark and Mary Stevens Neuroimaging and Informatics Institute (Stevens INI) have discovered that individuals experiencing significant physical limitations due to stroke may exhibit a "younger" brain structure in areas not directly affected by the injury. This unexpected phenomenon appears to be a testament to the brain’s capacity for self-reorganization and adaptation in the face of severe damage.
The research, a significant undertaking by the Enhancing NeuroImaging Genetics through Meta-Analysis (ENIGMA) Stroke Recovery Working Group, involved a comprehensive analysis of brain scans from over 500 stroke survivors. These invaluable data were meticulously collected from 34 distinct research centers spanning eight countries, creating an unprecedented global dataset. By deploying sophisticated deep learning models, which were trained on tens of thousands of magnetic resonance imaging (MRI) scans, the scientific team was able to estimate the "brain age" of specific regions within each hemisphere of the brain. This innovative approach allowed them to meticulously examine how stroke impacts both the structural integrity of the brain and the subsequent recovery process.
AI Unveils a Surprising Pattern of Brain Rewiring
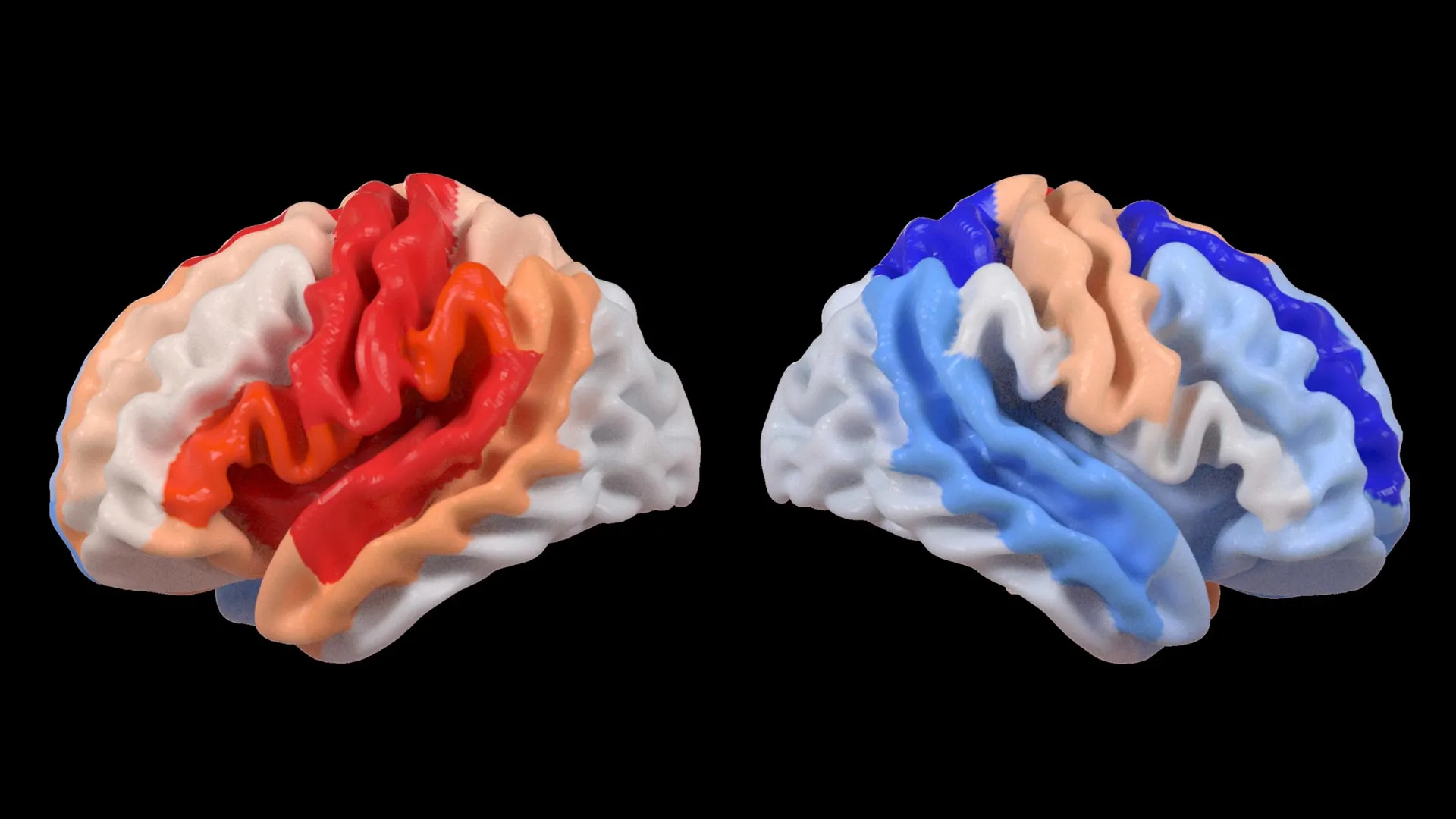
At the heart of this discovery lies the application of advanced artificial intelligence, specifically a type known as a graph convolutional network. This powerful AI system was instrumental in estimating the biological age of 18 distinct brain regions based on detailed MRI data. The researchers then compared these AI-generated "predicted brain ages" with each individual’s chronological age. The resulting metric, termed the brain-predicted age difference (brain-PAD), serves as a critical indicator of brain health and the extent of any deviation from expected aging patterns.
A crucial correlation emerged when these brain age measurements were juxtaposed with motor function scores. Stroke survivors who endured severe movement impairments, even after more than six months of dedicated rehabilitation, consistently displayed a younger-than-expected brain age in brain regions situated on the opposite side of the initial stroke. This effect was particularly pronounced within the frontoparietal network, a critical brain system involved in the intricate processes of movement planning, attention, and overall coordination.
"We were quite astonished by the findings," stated Hosung Kim, PhD, an associate professor of research neurology at the Keck School of Medicine of USC and a co-senior author of the study. "Our analysis revealed that while larger strokes demonstrably accelerate aging in the damaged hemisphere, they paradoxically cause the opposite side of the brain to appear younger. This striking pattern strongly suggests that the brain is actively reorganizing itself, essentially rejuvenating undamaged neural networks to compensate for the loss of function incurred by the stroke."
The Significance of Contralesional Neuroplasticity
The concept of contralesional neuroplasticity, the brain’s ability to reorganize and adapt in areas opposite to the site of injury, has been a subject of increasing interest in neuroscience. However, this new study provides compelling, large-scale evidence of this phenomenon in the context of stroke recovery, particularly in relation to severe motor deficits. The fact that undamaged regions exhibit signs of "youthful" brain structure in individuals with significant motor impairments points towards a dynamic compensatory mechanism.
Dr. Kim elaborated on this observation: "These findings indicate that when stroke damage leads to a greater loss of motor function, the undamaged regions on the opposite side of the brain may be actively adapting to help compensate. We observed this particularly in the contralesional frontoparietal network, which displayed a more ‘youthful’ pattern and is known to be crucial for motor planning, attention, and coordination. Rather than this pattern signifying a full recovery of movement, it may represent the brain’s sophisticated attempt to adjust and maintain function when the primary motor system can no longer operate normally. This offers us an entirely novel perspective on neuroplasticity, revealing insights that traditional imaging techniques might have missed."
Leveraging Big Data for Deeper Insights
The robustness of this study is significantly amplified by its reliance on the ENIGMA consortium. ENIGMA represents a global collaborative effort that aggregates data from over 50 countries, fostering a deeper, more nuanced understanding of the human brain across a wide spectrum of conditions. By meticulously standardizing MRI data and associated clinical information from numerous research groups, the team successfully constructed what is considered the largest stroke neuroimaging dataset of its kind.
"By pooling data from hundreds of stroke survivors worldwide and applying cutting-edge AI, we are able to detect subtle patterns of brain reorganization that would likely remain invisible in smaller, more localized studies," explained Arthur W. Toga, PhD, director of the Stevens INI and Provost Professor at USC. "These discoveries regarding regionally differential brain aging in chronic stroke hold immense potential to guide the development of personalized rehabilitation strategies in the future. The ability to identify these specific patterns of adaptation could revolutionize how we approach post-stroke care."
A Timeline of Discovery and Future Directions
The genesis of this research can be traced back to the ongoing efforts within the ENIGMA consortium to understand the neural underpinnings of various neurological conditions. The Stroke Recovery Working Group, formed to specifically address the complexities of stroke, began its data collection and collaborative efforts several years ago. The development and refinement of deep learning models capable of accurately estimating brain age from MRI scans, a process that has seen significant advancements in the last decade, provided the technological leap necessary for this particular study.
The data analysis phase, which involved the complex processing of hundreds of terabytes of MRI data, utilized significant computational resources and took several years to complete. The publication in The Lancet Digital Health marks the culmination of this extensive research endeavor.
Broader Implications for Stroke Rehabilitation
The implications of this study extend far beyond the academic realm, promising to reshape clinical approaches to stroke recovery. The ability to identify specific patterns of brain adaptation using AI could pave the way for highly personalized rehabilitation programs. Currently, stroke rehabilitation often follows standardized protocols. However, this research suggests that individual brains respond differently to injury, and understanding these unique adaptive mechanisms could lead to more effective and efficient therapeutic interventions.
For instance, identifying that specific undamaged regions are actively compensating could inform therapists about which areas to further stimulate or protect. Conversely, areas showing accelerated aging might require different therapeutic approaches. This personalized approach could lead to improved functional recovery, reduced long-term disability, and ultimately, an enhanced quality of life for stroke survivors.
The researchers are not resting on their laurels. Their future plans involve longitudinal studies, where they will follow stroke patients over extended periods, from the acute phase immediately following the stroke through their long-term recovery journey. By meticulously tracking how these brain aging patterns and structural changes evolve over time, clinicians may gain the ability to tailor treatments with unprecedented precision, adapting interventions to each individual’s unique recovery trajectory. The ultimate goal remains to optimize outcomes and significantly improve the lives of those affected by stroke.
Global Collaboration and Funding
The success of this ambitious research hinges on the collaborative spirit of the ENIGMA network. This global initiative, comprising researchers and institutions from around the world, provides the critical mass of data and expertise needed to tackle complex neurological questions. The standardization of data across diverse research centers is a testament to the dedication of its members.
The study received vital funding from the National Institutes of Health (NIH) under grant number R01 NS115845. Crucially, the research was also supported by a network of international collaborators from esteemed institutions, including the University of British Columbia, Monash University, Emory University, and the University of Oslo. This international cooperation underscores the global burden of stroke and the shared commitment to finding better solutions.
A visual explanation of the study’s findings on contralesional neuroplasticity and its association with motor impairment is available through a video produced by the Stevens INI, offering a valuable resource for both the scientific community and the general public seeking to understand this complex area of research.







