The landscape of Alzheimer’s disease treatment is on the cusp of a significant transformation, moving beyond therapies that merely slow progression to potentially reversing some of the underlying pathology. While current monoclonal antibody treatments have demonstrated the ability to modestly slow the disease’s relentless march, offering patients an average of 10 additional months of independence, these infusions, administered monthly or bi-monthly, represent a substantial logistical and financial commitment. Now, researchers at Washington University School of Medicine in St. Louis are pioneering an innovative cellular immunotherapy that utilizes genetically engineered brain cells, offering the tantalizing prospect of a single-injection treatment capable of both preventing and reducing amyloid plaque accumulation in the brain. This breakthrough, published on March 5 in the prestigious journal Science, represents a novel paradigm in the fight against neurodegenerative diseases.
The Dawn of a New Era in Alzheimer’s Treatment
For decades, the primary focus in Alzheimer’s drug development has been on targeting amyloid beta, a sticky protein that forms toxic plaques in the brain, a hallmark of the disease. The recent approval of monoclonal antibodies like aducanumab and lecanemab marked a pivotal moment, as these drugs were the first to demonstrate a statistically significant slowing of cognitive decline by clearing these amyloid plaques. However, these treatments are not without their challenges. Patients require regular intravenous infusions, often at specialized centers, and can experience side effects such as brain swelling and bleeding (ARIA – Amyloid-Related Imaging Abnormalities).
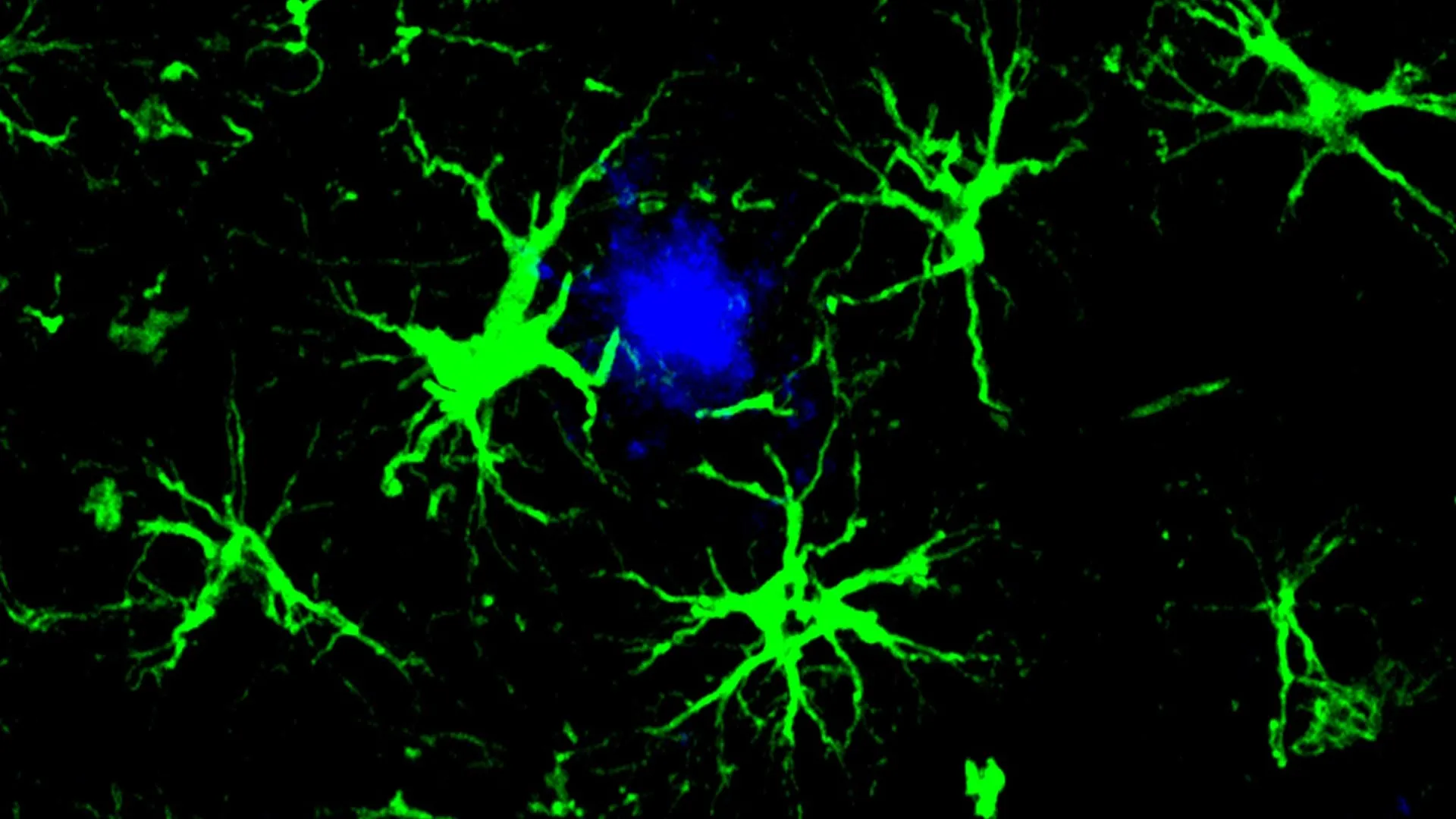
The Washington University study addresses these limitations by re-imagining the brain’s own cellular machinery. The researchers have engineered astrocytes, the most abundant glial cells in the brain, to act as potent amyloid-clearing agents. This approach draws inspiration from the success of CAR-T cell therapy in cancer treatment, where a patient’s own immune T cells are genetically modified to recognize and destroy cancer cells. In this Alzheimer’s adaptation, astrocytes are equipped with a Chimeric Antigen Receptor (CAR) – a synthetic receptor that allows them to specifically identify and bind to amyloid beta plaques.
Repurposing Brain Cells: The CAR-Astrocyte Revolution
The ingenious strategy involves genetically modifying astrocytes to express a CAR. These engineered cells, dubbed CAR-astrocytes, are designed to actively seek out and engulf amyloid beta proteins, effectively clearing them from the brain. "This study marks the first successful attempt at engineering astrocytes to specifically target and remove amyloid beta plaques in the brains of mice with Alzheimer’s disease," stated Dr. Marco Colonna, the Robert Rock Belliveau, MD, Professor of Pathology at WashU Medicine and senior author of the study. "Although more work needs to be done to optimize the approach and address potential side effects, these results open up an exciting new opportunity to develop CAR-astrocytes into an immunotherapy for neurodegenerative diseases and even brain tumors."
The genesis of this research lies in understanding the fundamental pathology of Alzheimer’s disease. It begins with the aberrant accumulation of amyloid beta peptides, which aggregate into oligomers and eventually form the characteristic plaques. These plaques are believed to initiate a cascade of neuroinflammatory and neurotoxic events, leading to synaptic dysfunction, neuronal loss, and the cognitive decline that defines Alzheimer’s. While the brain’s resident immune cells, microglia, normally play a role in clearing cellular debris, they can become overwhelmed and dysfunctional in the face of widespread amyloid pathology.
A Timeline of Discovery and Innovation
The journey leading to this groundbreaking discovery involved years of meticulous research into the brain’s cellular ecosystem and the mechanisms of amyloid deposition. The Washington University team, led by Dr. Yun Chen, a postdoctoral researcher in the laboratory of Dr. David M. Holtzman, a leading neurologist in Alzheimer’s research, focused on astrocytes due to their abundance and supportive role in neuronal function. Astrocytes are typically involved in maintaining the brain’s environment, regulating neurotransmitter levels, and providing metabolic support to neurons. The hypothesis was that by equipping these versatile cells with a specific targeting mechanism, they could be repurposed for a more aggressive cleanup operation against amyloid.
The researchers utilized a harmless viral vector to introduce the gene encoding the CAR into astrocytes within living mice. This gene-directed approach ensures that the CAR is expressed on the surface of the astrocytes, enabling them to recognize and bind to amyloid beta. Once bound, the CAR-astrocytes are designed to internalize and break down the amyloid proteins.
Pre-clinical Trials: Promising Results in Mouse Models
The efficacy of the CAR-astrocyte therapy was rigorously tested in genetically engineered mice that are predisposed to developing amyloid plaques, mimicking aspects of human Alzheimer’s disease. Two distinct experimental arms were established:
- Prophylactic Treatment: A group of younger mice received the CAR-astrocyte therapy before the onset of significant amyloid plaque formation. This arm aimed to assess the therapy’s ability to prevent the disease from taking hold.
- Therapeutic Treatment: An older group of mice, whose brains already harbored substantial amyloid plaques, received the CAR-astrocyte therapy. This arm evaluated the treatment’s capacity to reduce existing pathology.
The results were compelling. In the prophylactic group, the CAR-astrocyte treatment effectively prevented the formation of amyloid plaques. By approximately six months of age, a time when untreated control mice exhibited widespread plaque accumulation, the treated mice showed no detectable plaques. This suggests a potent preventative capability.
In the therapeutic group, which began treatment with established plaques, the CAR-astrocyte therapy demonstrated a significant reduction in amyloid burden. Compared to control mice that received a virus without the CAR gene, the treated mice exhibited a reduction in amyloid plaque levels by approximately 50 percent after three months. This substantial clearance of existing plaques is a critical finding, indicating that the therapy can actively reverse pathology, not just prevent its progression.
The Single-Injection Advantage: A Paradigm Shift
The most revolutionary aspect of this research, and a key differentiator from current antibody therapies, is the potential for a single-injection administration. While monoclonal antibodies require ongoing infusions, the CAR-astrocyte therapy, by genetically engineering the brain’s own cells, offers the possibility of a long-lasting therapeutic effect from a single administration. This could drastically reduce the burden on patients and healthcare systems, making advanced Alzheimer’s treatment more accessible and manageable.
Dr. David M. Holtzman, the Barbara Burton and Reuben M. Morriss III Distinguished Professor of Neurology at WashU Medicine and a co-author on the study, highlighted this crucial advantage: "Consistent with the antibody drug treatments, this new CAR-astrocyte immunotherapy is more effective when given in the earlier stages of the disease. But where it differs, and where it could make a difference in clinical care, is in the single injection that successfully reduced the amount of harmful brain proteins in mice."
Implications and Future Directions
The implications of this research extend far beyond Alzheimer’s disease. The CAR-astrocyte technology holds immense potential for treating a spectrum of neurodegenerative disorders characterized by protein aggregation, such as Parkinson’s disease and Frontotemporal Dementia. Furthermore, the underlying principle of engineering glial cells for therapeutic purposes could be adapted to target other diseases affecting the central nervous system.
The research team is already looking ahead, focusing on refining the CAR-astrocyte therapy. Future work will prioritize enhancing the precision with which these engineered cells target harmful proteins, ensuring minimal disruption to normal brain cell function. The development of strategies to precisely control the duration and intensity of the CAR-astrocyte activity will also be crucial for optimizing safety and efficacy in human trials.
Moreover, the researchers envision adapting this technology for the treatment of brain tumors. By re-engineering the CAR homing device to recognize specific markers present on cancer cells, astrocytes could be redirected to actively seek out and destroy tumor cells, offering a novel approach to combating some of the most aggressive forms of cancer.
The Washington University team has filed a patent for their CAR-astrocyte engineering method, signaling a commitment to translating this promising pre-clinical work into tangible clinical applications. While human trials are still some way off, the successful demonstration of this single-injection, cell-based immunotherapy in mice represents a significant leap forward, offering renewed hope in the ongoing battle against devastating neurological diseases. The scientific community will be keenly watching the progress of this innovative approach as it navigates the complex path from laboratory bench to patient bedside.