A groundbreaking new study published in The Lancet Digital Health is shedding new light on the remarkable adaptability of the human brain following a stroke. Researchers from the USC Mark and Mary Stevens Neuroimaging and Informatics Institute (Stevens INI) have uncovered compelling evidence that suggests the brain can engage in a surprising form of self-preservation and reorganization, even in the face of severe injury. The findings indicate that individuals experiencing significant physical impairments after a stroke may exhibit a "younger" brain structure in areas not directly affected by the damage, a phenomenon that appears to be a testament to the brain’s innate capacity for adaptation and functional compensation.
This pioneering research was conducted as an integral part of the Enhancing NeuroImaging Genetics through Meta-Analysis (ENIGMA) Stroke Recovery Working Group, a global collaborative effort aimed at understanding the complex interplay between genetics, neuroimaging, and stroke recovery. The scientific team meticulously analyzed brain scans from over 500 stroke survivors, with the data originating from 34 distinct research centers spanning eight different countries. By employing sophisticated deep learning models, which were trained on an extensive dataset comprising tens of thousands of Magnetic Resonance Imaging (MRI) scans, the researchers were able to estimate the "brain age" of various regions within each hemisphere of the brain. This analysis allowed them to examine how stroke impacts both the structural integrity of the brain and the subsequent recovery process.
The implications of this research are profound, offering a novel perspective on neuroplasticity—the brain’s ability to reorganize itself by forming new neural connections throughout life. Traditionally, neuroplasticity has been observed as the brain’s capacity to learn new skills or recover lost function. However, this study introduces a new dimension by quantifying this adaptation through the lens of "brain aging."
AI-Powered Insights into Brain Rewiring
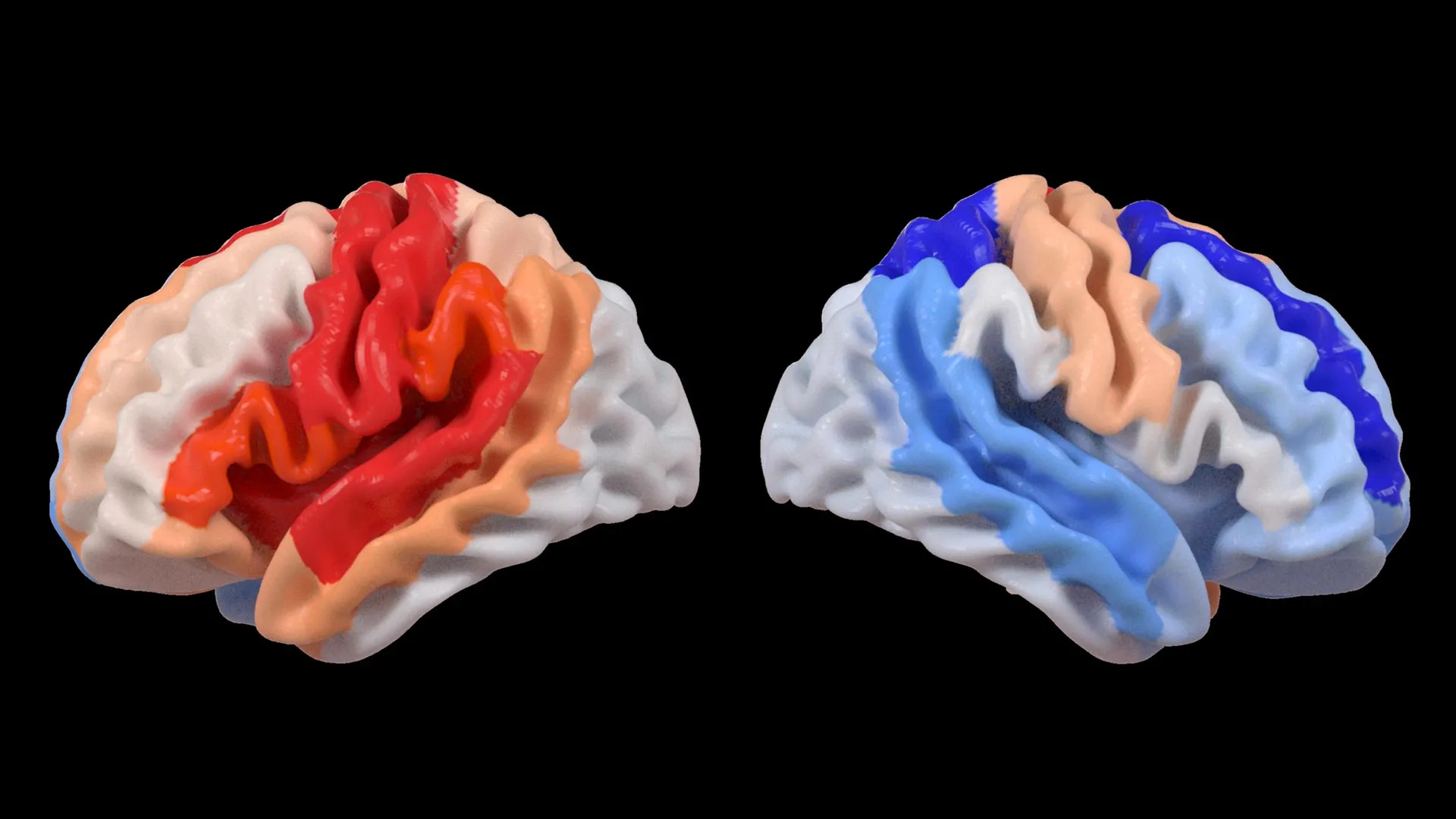
At the heart of this investigation lies the innovative application of artificial intelligence, specifically a type of machine learning known as a graph convolutional network. This advanced AI system was instrumental in estimating the biological age of 18 specific brain regions based on the detailed information captured by MRI scans. The estimated age for each region was then compared to the individual’s chronological age. This comparison yielded a metric known as the brain-predicted age difference (brain-PAD), a value that serves as a crucial indicator of overall brain health and integrity. A significant positive brain-PAD, where the predicted age is younger than the actual age, is generally associated with better brain health, while a negative brain-PAD suggests accelerated aging in that region.
The research team then correlated these sophisticated brain age measurements with objective assessments of motor function, the ability to move and perform physical tasks. A striking and consistent pattern emerged from this analysis. Stroke survivors who exhibited severe impairments in movement, even after enduring more than six months of intensive rehabilitation therapy, consistently displayed a younger-than-expected brain age in regions located on the opposite side of the brain from the initial stroke. This effect was particularly pronounced in the frontoparietal network, a critical brain system responsible for a range of executive functions including movement planning, attention, and the coordination of complex actions.
Dr. Hosung Kim, an associate professor of research neurology at the Keck School of Medicine of USC and a co-senior author of the study, elaborated on these findings. "We discovered that larger strokes tend to accelerate aging in the damaged hemisphere, but paradoxically, they make the opposite side of the brain appear younger," Dr. Kim stated. "This distinct pattern strongly suggests that the brain may be actively reorganizing itself. In essence, it appears to be rejuvenating undamaged networks to compensate for the functions that have been lost due to the injury."
This observation offers a compelling explanation for why some individuals might show resilience or partial recovery of function despite significant initial damage. The brain, faced with the challenge of diminished capacity in one hemisphere, appears to be proactively bolstering the capabilities of the contralateral (opposite) hemisphere.
The Power of Large-Scale Data and Global Collaboration
The success of this study is intrinsically linked to the robust infrastructure and data-sharing capabilities of the ENIGMA project. ENIGMA operates as a vast global collaboration, bringing together researchers and data from over 50 countries to foster a deeper, more comprehensive understanding of the human brain across a wide spectrum of neurological conditions. By meticulously standardizing MRI data and associated clinical information from numerous research groups, the ENIGMA Stroke Recovery Working Group was able to construct an unprecedentedly large and diverse stroke neuroimaging dataset. This scale of data is crucial for identifying subtle patterns and effects that might be missed in smaller, single-institution studies.
Arthur W. Toga, PhD, director of the Stevens INI and Provost Professor at USC, highlighted the significance of this collaborative approach. "By pooling data from hundreds of stroke survivors worldwide and applying cutting-edge AI, we are able to detect subtle patterns of brain reorganization that would simply be invisible in smaller studies," Dr. Toga explained. "These findings, which reveal regionally differential brain aging in chronic stroke, hold the potential to guide the development of highly personalized rehabilitation strategies in the future."
The implications of this large-scale data analysis extend beyond simply identifying a phenomenon. It provides a foundation for understanding the heterogeneity of stroke recovery. Factors such as the size and location of the stroke, the age of the individual, pre-existing brain health, and even genetic predispositions can all influence how the brain adapts. The ENIGMA initiative, by encompassing such a broad range of participants and data, allows for a more nuanced exploration of these contributing factors.
A Timeline of Discovery and Future Directions
The journey leading to these findings can be traced back to the broader ENIGMA project’s inception, which has been systematically building its comprehensive neuroimaging databases for years. The specific focus on stroke recovery intensified as researchers recognized the potential of advanced computational methods to unlock hidden insights within existing data.
Early Stages of ENIGMA and Stroke Research (circa 2010s): The ENIGMA consortium began aggregating neuroimaging data for various neurological and psychiatric conditions, establishing standardized protocols for data acquisition and sharing. Stroke was identified as a key area for investigation due to its significant public health impact and the inherent complexity of brain recovery.
Development of Deep Learning Models (Mid-2010s onwards): Advances in artificial intelligence, particularly in deep learning, created new possibilities for analyzing complex neuroimaging data. Researchers began developing and refining AI models capable of identifying subtle structural and functional patterns within brain scans.
Formation of the ENIGMA Stroke Recovery Working Group (Late 2010s): This specialized working group was established to consolidate efforts and data specifically focused on understanding the mechanisms of stroke recovery. The goal was to leverage the collective power of ENIGMA’s global network.
Data Collection and Initial Analysis (Early 2020s): The 500+ stroke survivor scans were collected and standardized. The deep learning models were trained and applied to estimate brain age across various regions.
Identification of the "Younger" Contralesional Hemisphere Phenomenon (2023-2024): The correlation between severe motor impairment and younger brain age in the unaffected hemisphere was systematically identified and validated. This marked the core discovery of the study.
Publication in The Lancet Digital Health (Present): The findings were formally published, bringing this significant discovery to the wider scientific and medical communities.
Looking ahead, the researchers are keenly focused on translating these insights into tangible improvements for stroke survivors. The next phase of their work involves longitudinally tracking patients from the immediate aftermath of a stroke through their long-term recovery trajectories. By observing how brain aging patterns and structural changes evolve over time, clinicians may gain the ability to tailor therapeutic interventions to each individual’s unique recovery process. This personalized approach holds the promise of optimizing treatment strategies, ultimately leading to enhanced recovery outcomes and a significant improvement in the quality of life for millions affected by stroke worldwide.
Broader Impact and Implications for Stroke Rehabilitation
The findings have significant implications for how stroke rehabilitation is conceived and delivered. The identification of a "younger" and potentially more adaptable contralateral hemisphere in severely impaired individuals suggests that therapeutic strategies could be designed to more effectively harness the compensatory potential of these undamaged brain regions. This could involve targeted stimulation techniques or specialized exercises aimed at enhancing the connectivity and function of these brain areas.
Furthermore, the brain-PAD metric, derived from AI analysis, could serve as a valuable biomarker for predicting recovery potential and monitoring treatment effectiveness. Instead of relying solely on traditional clinical assessments, which can sometimes be slow to reflect subtle changes, brain-PAD could offer a more objective and sensitive measure of the brain’s ongoing adaptive processes. This could allow for earlier adjustments to rehabilitation plans, ensuring that patients receive the most appropriate interventions at the right time.
The study also underscores the increasing importance of large-scale, multi-institutional collaborations like ENIGMA. By pooling resources and data, researchers can tackle complex scientific questions that would be insurmountable for individual labs. This model of collaborative science is becoming increasingly vital in addressing global health challenges.
While the findings are promising, it is important to acknowledge that this is a step in an ongoing scientific journey. Further research is needed to fully elucidate the precise mechanisms underlying this contralateral rejuvenation and to determine its direct causal link to functional recovery. However, the current study provides a powerful new lens through which to view the brain’s remarkable resilience and adaptive capacity in the wake of devastating injury.
The potential for personalized stroke recovery is immense. By understanding the intricate ways the brain reorganizes itself, scientists and clinicians can move closer to developing therapies that are not one-size-fits-all, but rather precisely tailored to the individual needs and adaptive capabilities of each stroke survivor. This research represents a significant stride toward that future, offering hope for improved outcomes and a better quality of life for those navigating the challenges of post-stroke recovery.
For those interested in delving deeper into the fascinating relationship between contralesional neuroplasticity and motor impairment following a stroke, the Stevens INI has produced an informative video that can be accessed via a provided link.
The study, formally titled "Deep learning prediction of MRI-based regional brain age reveals contralesional neuroplasticity associated with severe motor impairment in chronic stroke: A worldwide ENIGMA study," received crucial funding from the National Institutes of Health (NIH) under grant R01 NS115845. Its international reach was bolstered by vital support from collaborators at esteemed institutions including the University of British Columbia, Monash University, Emory University, and the University of Oslo, exemplifying the global nature of modern scientific inquiry.







