The landscape of Alzheimer’s disease treatment is on the cusp of a significant transformation, moving beyond therapies that merely slow the progression of this devastating neurodegenerative condition. While recent advancements in monoclonal antibody treatments have offered a beacon of hope, extending patient independence by an average of ten months through the reduction of harmful amyloid protein buildup, a groundbreaking cellular immunotherapy developed by researchers at Washington University School of Medicine in St. Louis is now offering the tantalizing prospect of a single-injection therapy. This innovative approach, detailed in a landmark publication on March 5 in the prestigious journal Science, demonstrates the potential to prevent or significantly reduce amyloid plaques, the pathological hallmarks of Alzheimer’s disease, with unprecedented efficacy and convenience.
The current generation of Alzheimer’s medications, primarily monoclonal antibodies, target amyloid-beta, a sticky protein that aggregates in the brain, forming plaques that disrupt neuronal function and ultimately lead to cognitive decline and brain atrophy. These treatments, while a crucial step forward, require frequent, large-dose infusions, often administered intravenously once or twice a month. This regimen presents a logistical and financial burden for patients and healthcare systems alike, and the benefits, though meaningful, are often realized over a protracted period. Scientists have long sought more sustainable and potent therapeutic strategies, and the work emerging from Washington University may represent a paradigm shift.
Repurposing Brain Cells for Amyloid Clearance: A CAR-T Inspired Innovation
The new experimental therapy draws inspiration from the highly successful chimeric antigen receptor (CAR)-T cell therapy, a revolutionary treatment for certain blood cancers. In CAR-T therapy, a patient’s own T cells are genetically engineered in a laboratory to recognize and attack cancer cells. This Alzheimer’s approach adapts this concept by engineering a different type of brain cell: astrocytes.
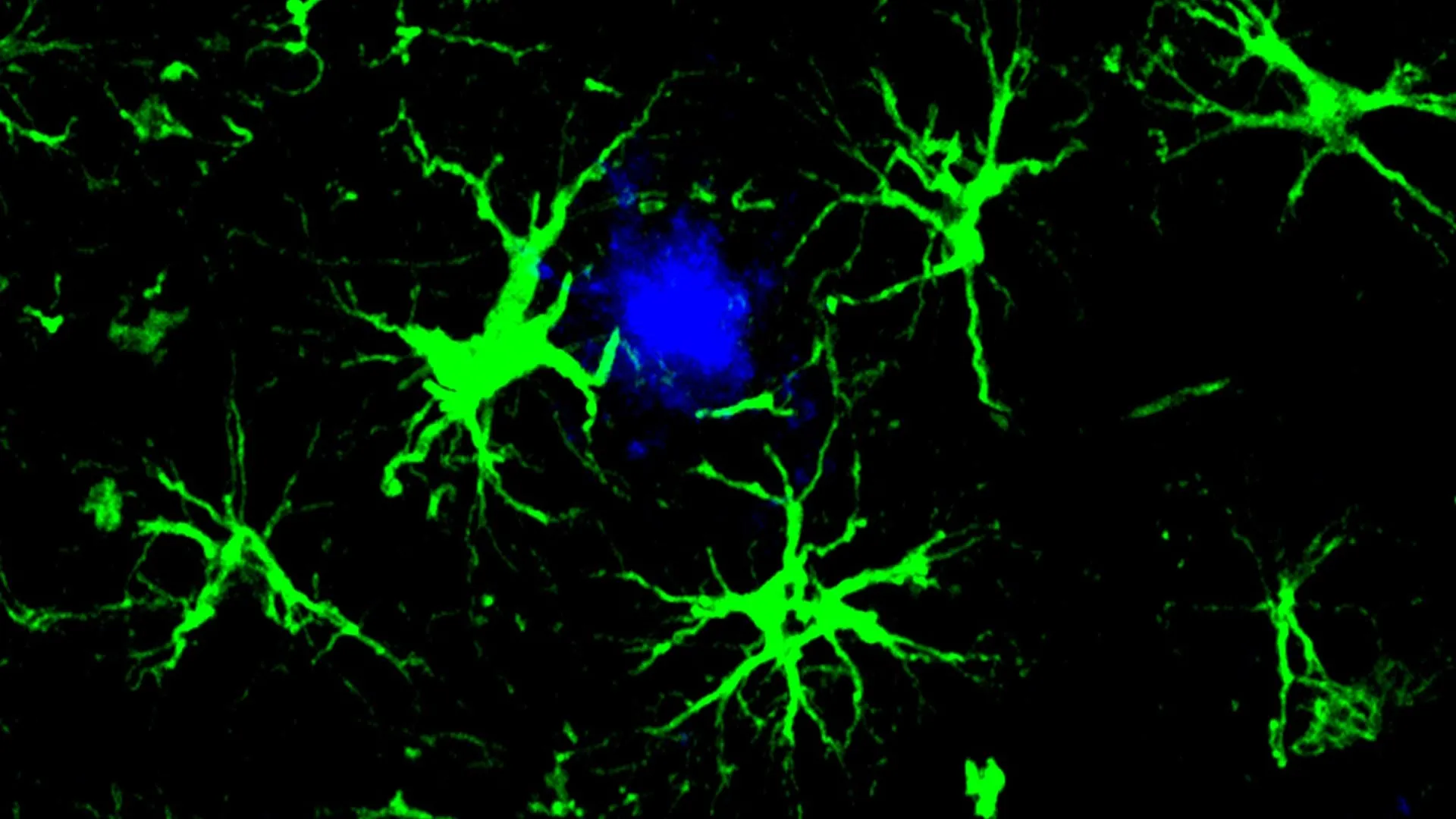
Astrocytes, the most abundant glial cells in the central nervous system, play vital roles in supporting neuronal health, regulating synaptic function, and maintaining the brain’s internal environment. In this novel strategy, researchers modified astrocytes by equipping them with a CAR "homing device." This engineered component allows the astrocytes to specifically identify and bind to amyloid-beta plaques. Once attached, these reprogrammed astrocytes act as highly effective cellular "clean-up crews," actively engulfing and degrading the harmful protein aggregates.
"This study marks the first successful attempt at engineering astrocytes to specifically target and remove amyloid beta plaques in the brains of mice with Alzheimer’s disease," stated Dr. Marco Colonna, the Robert Rock Belliveau, MD, Professor of Pathology at WashU Medicine and the study’s senior author. "Although more work needs to be done to optimize the approach and address potential side effects, these results open up an exciting new opportunity to develop CAR-astrocytes into an immunotherapy for neurodegenerative diseases and even brain tumors."
Understanding the Devastation of Amyloid Plaques
Alzheimer’s disease, a progressive neurodegenerative disorder affecting millions worldwide, is characterized by the insidious accumulation of amyloid-beta peptides. These peptides misfold and aggregate, forming insoluble plaques that deposit in the spaces between neurons. This process triggers a cascade of pathological events, including inflammation, oxidative stress, and the formation of tau tangles, which further impair neuronal communication and survival. Ultimately, these disruptions lead to a gradual loss of brain mass, particularly in regions critical for memory, cognition, and behavior, resulting in the debilitating symptoms of Alzheimer’s.
Under normal circumstances, the brain possesses its own sophisticated waste removal system, primarily managed by microglia, the resident immune cells of the central nervous system. Microglia are responsible for clearing cellular debris, misfolded proteins, and other harmful substances. However, in the context of neurodegenerative diseases like Alzheimer’s, these protective mechanisms can become overwhelmed. The chronic presence of amyloid plaques can lead to microglial dysfunction, rendering them less effective at clearing the accumulating pathology and potentially contributing to neuroinflammation, further exacerbating neuronal damage.
The Genesis of the CAR-Astrocyte Therapy: A Timeline of Innovation
The research leading to this significant breakthrough began with the recognition that astrocytes, despite their supportive roles, could potentially be harnessed for therapeutic intervention. Dr. Yun Chen, the first author of the study and a postdoctoral researcher in the lab of Dr. David M. Holtzman, the Barbara Burton and Reuben M. Morriss III Distinguished Professor of Neurology at WashU Medicine, spearheaded the effort to redesign astrocytes.
The team employed a harmless virus to introduce a gene encoding a CAR into astrocytes. This genetic modification effectively reprogrammed the astrocytes, imbuing them with the ability to recognize and bind to amyloid-beta proteins. Once the CAR receptor is expressed on the astrocyte’s surface, the cell gains the capacity to actively engulf and degrade the amyloid-beta.
The researchers then tested this novel CAR-astrocyte therapy in genetically engineered mice that are predisposed to developing amyloid plaques, mimicking key aspects of human Alzheimer’s disease. These mice begin accumulating plaques around six months of age. The study strategically divided these mice into two key experimental groups:
- Preventative Group: This group received the CAR gene therapy before the onset of amyloid plaque formation. The aim was to assess the therapy’s ability to prevent the pathological process from initiating.
- Therapeutic Group: This group received the treatment after significant amyloid plaque buildup had already occurred in their brains. This group was designed to evaluate the therapy’s efficacy in reducing existing pathology.
After a three-month observation period, the results were compelling. In the younger mice that received the CAR gene therapy prophylactically, the development of amyloid plaques was effectively halted. By the age of six months, when their untreated counterparts exhibited brains heavily laden with plaques, the treated mice showed no detectable signs of plaque accumulation. This suggests a powerful preventative capability for the CAR-astrocyte therapy.
In the older mice that already had established plaques, the CAR-astrocyte treatment demonstrated a remarkable capacity to reduce existing amyloid burden. The researchers observed a significant reduction in amyloid plaque levels, approximately 50 percent, when compared to control mice that received a virus lacking the CAR gene. This finding highlights the therapy’s potential to reverse or significantly mitigate ongoing disease processes.
A Single Injection Solution: Redefining Treatment Modalities
The implications of a single-injection therapy for Alzheimer’s disease are profound. The current reliance on intravenous infusions necessitates regular clinical visits, potentially leading to patient fatigue, anxiety, and increased healthcare costs. A single, targeted injection could drastically simplify treatment administration, improve patient compliance, and potentially reduce the overall economic burden associated with long-term Alzheimer’s care.
"Consistent with the antibody drug treatments, this new CAR-astrocyte immunotherapy is more effective when given in the earlier stages of the disease," noted Dr. David M. Holtzman, a co-author on the study. "But where it differs, and where it could make a difference in clinical care, is in the single injection that successfully reduced the amount of harmful brain proteins in mice."
The researchers have already taken steps to protect their innovation, filing a patent for their CAR-astrocyte engineering method through the Office of Technology Management at Washington University. This proactive measure underscores the potential commercial and clinical significance of their discovery.
Broader Implications: A Platform for Neurological and Oncological Therapies
The success of this CAR-astrocyte approach extends beyond Alzheimer’s disease. The underlying principle of engineering glial cells to target specific pathological markers holds immense promise for a wide range of neurological disorders. Researchers are actively exploring the possibility of adapting this technology to target other neurodegenerative conditions characterized by protein misfolding and aggregation, such as Parkinson’s disease or amyotrophic lateral sclerosis (ALS).
Furthermore, the research team envisions a future where this CAR-astrocyte technology can be repurposed to combat brain tumors. By modifying the CAR homing device to recognize specific surface markers present on cancer cells, astrocytes could be redirected from their plaque-clearing duties to actively seek out and destroy tumor cells. This innovative strategy could pave the way for novel immunotherapies for challenging conditions like glioblastoma, offering new hope to patients with limited treatment options.
The path from laboratory discovery to clinical application is often long and complex, involving rigorous preclinical testing, extensive safety evaluations, and multiple phases of human clinical trials. However, the findings published in Science represent a significant leap forward in the quest for effective Alzheimer’s treatments. The development of a potential single-injection cellular immunotherapy that can prevent or reduce amyloid pathology marks a pivotal moment, offering a glimpse into a future where Alzheimer’s disease may be not only managed but potentially prevented or even reversed. The scientific community will be keenly watching as this promising technology continues to evolve, with the hope that it will ultimately translate into tangible benefits for patients worldwide.







