Researchers at the University of Illinois Urbana-Champaign have unveiled a groundbreaking method for dissecting the intricate wiring of the brain, a development poised to revolutionize neuroscience. This novel technique, dubbed Connectome-seq, employs molecular "barcodes" to tag individual neurons, enabling scientists to map neural connections with unparalleled speed and precision. Initial applications of Connectome-seq have already yielded a detailed map of thousands of neural connections within the mouse brain, offering a powerful new lens through which to understand brain function, development, and the origins of neurological disorders.
The Challenge of Mapping the Brain’s Intricate Network
For decades, unraveling the complex circuitry of the brain has been a formidable challenge. The sheer density of connections, coupled with the delicate nature of neural tissue, has historically made mapping a laborious and time-consuming endeavor. Traditional methods often involved meticulously slicing brain tissue into infinitesimally thin sections, a process akin to dissecting a living organism into microscopic fragments. These slices were then painstakingly imaged under powerful microscopes, and the resulting two-dimensional images were manually pieced together to reconstruct three-dimensional neural pathways. This analog approach, while yielding foundational insights, was inherently limited in its scale and resolution, making it impractical for comprehensively charting vast neural networks.
More recent advancements have introduced sequencing-based tools capable of labeling numerous neurons simultaneously. However, these methods typically indicate the broad reach of a neuron, showing where its extensions, or axons, project within the brain. They often fall short of identifying the precise cellular targets with which a neuron forms functional connections – the synapses. Understanding these synaptic connections is crucial, as it is at these junctions that information is transmitted from one neuron to another, forming the basis of all thought, emotion, and behavior. The absence of this fine-grained synaptic resolution has represented a significant bottleneck in neuroscience research, hindering a complete understanding of how neural circuits operate and how their dysfunction contributes to disease.
Connectome-seq: A Paradigm Shift in Neural Mapping
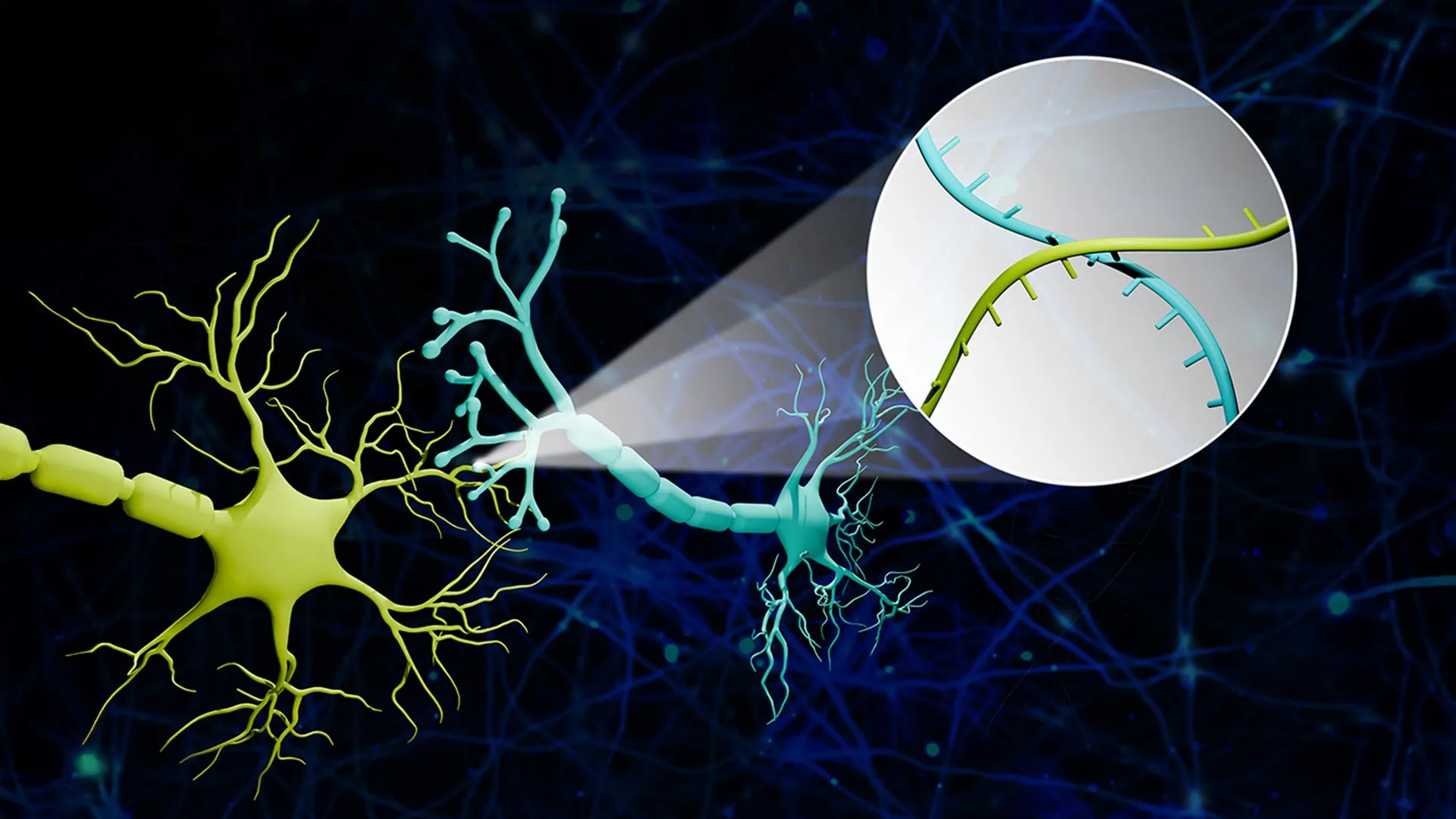
The Connectome-seq platform, developed by the team led by Professor Boxuan Zhao of the University of Illinois Urbana-Champaign’s Department of Cell and Developmental Biology, directly addresses these limitations. The core innovation lies in the ingenious use of RNA molecules as unique molecular identifiers, or "barcodes," for each neuron. These specialized barcodes are synthesized within the main body of a neuron and are then meticulously transported along the neuron’s axon to its synaptic terminals. When two neurons connect at a synapse, their respective barcodes are brought into close proximity.
The subsequent stage of the Connectome-seq process involves the isolation of these synaptic junctions. High-throughput sequencing technology is then employed to read the barcodes present at each isolated synapse. By identifying which pairs of barcodes are found together, researchers can definitively determine which neurons are directly connected. This effectively translates the complex problem of neural connectivity into a manageable sequencing challenge, allowing for the reconstruction of neural networks on an unprecedented scale.
Professor Zhao elaborated on this innovative approach, drawing an analogy to illustrate its elegance: "We translated the neural connectivity problem into a sequencing problem. Imagine a big bunch of balloons. The main body of each balloon has its unique barcode stickers all over it, and some move down to the end of the string. If two balloons are tied together at the end, the two barcodes meet at the junction. Then we snip out the knots and sequence the barcodes in each one. If the same knot has stickers from balloon A and balloon B, we know these two balloons are tied together. We are doing this in the brain, just on the level of thousands of neuron cells. With this information, we can reconstruct a sophisticated map that represents the connections among all these seemingly floaty groups." This analogy highlights the method’s ability to link individual components (neurons) based on their co-occurrence at functional points (synapses).
Accelerating Discovery: Mapping the Pontocerebellar Circuit
The efficacy of Connectome-seq was demonstrated in a pilot study where the researchers applied the technique to map a specific neural circuit in the mouse brain known as the pontocerebellar circuit. This circuit plays a vital role in motor control and coordination, linking the pons, a brainstem structure, with the cerebellum. The analysis, which involved mapping over 1,000 neurons, revealed previously uncharacterized patterns of connectivity. Notably, the study uncovered direct synaptic links between cell types that had not previously been known to communicate directly in the adult brain. This discovery underscores the power of Connectome-seq to uncover hidden or overlooked neural relationships that could be critical for understanding circuit function.
The implications of these findings are substantial. The pontocerebellar circuit is a relatively well-studied area, and the fact that Connectome-seq could identify novel connections within it suggests its immense potential for exploring more complex and less understood brain regions. "With improvements already underway in our lab, we are confident that we can make it even better and eventually reach the goal of mapping the whole mouse brain," Professor Zhao stated, expressing optimism about the future scalability and refinement of the technology. The current mapping of thousands of connections represents a significant leap from previous capabilities, but the ultimate goal of a comprehensive connectome of an entire organism, or even a significant portion of a mammalian brain, is now within clearer view.
Historical Context: The Evolution of Connectomics
The quest to map the brain’s connections, often referred to as "connectomics," is not a new one. Early efforts in the field were primarily driven by electron microscopy, a technique that allowed for high-resolution imaging of neural ultrastructure. However, the sheer volume of data generated by electron microscopy and the manual effort required for reconstruction made it impossible to map even small sections of the brain at synaptic resolution within a reasonable timeframe. For instance, mapping a cubic millimeter of the fruit fly brain, a relatively simple nervous system, took years of dedicated effort.
The advent of genetic tools and advanced imaging techniques in the early 2000s began to accelerate progress. Methods like viral tracing allowed researchers to track the pathways of neuronal projections, providing a broader overview of connectivity. Later, genetically encoded fluorescent proteins enabled the visualization of entire neuronal populations. Yet, the challenge of achieving synaptic resolution on a large scale persisted. Sequencing-based approaches, which emerged more recently, offered a promising avenue for high-throughput analysis, but the initial iterations often lacked the precision to identify individual synaptic partners. Connectome-seq appears to bridge this gap, combining the scalability of sequencing with the critical need for synaptic-level accuracy.
The Promise for Neurological Disorders and Therapeutic Development
The potential impact of Connectome-seq extends far beyond fundamental neuroscience research. Its speed, scalability, and high resolution make it an invaluable tool for investigating the intricate neural underpinnings of neurological and psychiatric disorders. Conditions such as Alzheimer’s disease, Parkinson’s disease, schizophrenia, and autism spectrum disorder are all characterized by disruptions in neural circuit function. By enabling scientists to rapidly and precisely map these circuits, Connectome-seq offers a pathway to understanding how these diseases manifest at the network level.
"When engineering a computer, you need to know the circuitry of the central processing unit. If you don’t know how everything is wired together, you can’t understand its function, optimize it or fix it when something breaks. We are approaching the brain the same way," Professor Zhao emphasized, drawing a powerful analogy to engineering principles. This perspective highlights the fundamental importance of understanding the brain’s "wiring diagram" for both basic comprehension and practical application.
The ability to compare the connectomes of healthy brains with those affected by disease at various stages is a key advantage. This comparative analysis could reveal subtle yet critical alterations in synaptic connections that precede the onset of overt symptoms. For conditions like Alzheimer’s disease, where a cascade of pathological events leads to neuronal dysfunction and death, identifying the earliest points of circuit breakdown could be transformative.
"With sequencing-based approaches, the time and cost are greatly reduced, which really makes it possible to see differences in different brains," Zhao explained. "We could see where connections change, where the most vulnerable parts of the brain are, perhaps before symptoms even appear. For example, if we can catch where exactly the weak link is that kick starts the whole catastrophic cascade in Alzheimer’s disease, can we specifically strengthen those connections to where the disease slows or does not progress?" This points towards the exciting prospect of developing circuit-guided therapeutic interventions. By precisely identifying faulty connections, future treatments could be designed to repair, reroute, or reinforce specific neural pathways, offering a more targeted and potentially more effective approach to treating complex brain disorders.
Future Directions and Broader Implications
The research, published in the prestigious journal Nature Methods, has garnered significant attention within the scientific community. While Connectome-seq has demonstrated remarkable capabilities, the researchers are already focused on further refinements and expansions. Efforts are underway to increase the throughput even further, aiming to map larger volumes of brain tissue and ultimately achieve whole-brain connectivity maps. The development of more sophisticated computational tools for analyzing the massive datasets generated by Connectome-seq will also be crucial.
The implications of this technology are far-reaching. Beyond disease research, Connectome-seq could accelerate our understanding of learning and memory, the development of cognitive abilities, and the neural basis of consciousness. It offers a powerful tool for investigating how environmental factors and experiences shape brain circuitry throughout life. Furthermore, as artificial intelligence continues to advance, understanding the biological principles of efficient and robust neural computation, as revealed by detailed connectomic maps, could inspire new paradigms in machine learning and AI design.
The research was supported by significant funding from several prominent institutions, including a Neuro-omics Initiative grant from the Wu Tsai Neurosciences Institute of Stanford University, as well as grants from the Elsa U. Pardee Foundation and the Edward Mallinckrodt Jr. Foundation. This multidisciplinary support underscores the perceived importance and potential impact of this work in advancing brain science.
In conclusion, the development of Connectome-seq marks a pivotal moment in neuroscience. By providing a fast, detailed, and scalable method for mapping neural connections at the synaptic level, this technology promises to unlock new frontiers in our understanding of the brain, paving the way for significant advancements in the diagnosis and treatment of neurological disorders. The era of truly comprehensive brain mapping has arrived, offering hope for unraveling the mysteries of the mind and alleviating the burden of devastating brain diseases.







