A groundbreaking study published in The Lancet Digital Health has unveiled a surprising capacity of the human brain to adapt following a stroke, even in cases of severe physical impairment. Researchers at the USC Mark and Mary Stevens Neuroimaging and Informatics Institute (Stevens INI), a leading hub for neuroscience research, have identified a phenomenon where undamaged areas of the brain in stroke survivors may exhibit structural characteristics associated with a younger biological age. This intriguing finding suggests a remarkable inherent ability of the brain to reorganize and compensate for lost function, a concept central to neuroplasticity.
The research, a significant undertaking by the Enhancing NeuroImaging Genetics through Meta-Analysis (ENIGMA) Stroke Recovery Working Group, leveraged an unprecedented scale of data. Scientists meticulously analyzed brain scans from over 500 individuals who had experienced a stroke. These crucial imaging data were collected from 34 different research centers spanning eight countries, creating a robust and diverse dataset. To unlock the subtle patterns within this vast collection, the team employed sophisticated deep learning models. These artificial intelligence systems were trained on tens of thousands of magnetic resonance imaging (MRI) scans, enabling them to estimate the "brain age" of specific regions within each hemisphere of the brain. This allowed researchers to meticulously examine how stroke impacts both the structural integrity of the brain and the subsequent recovery process.
AI Uncovers a Paradoxical Youthfulness in the Injured Brain
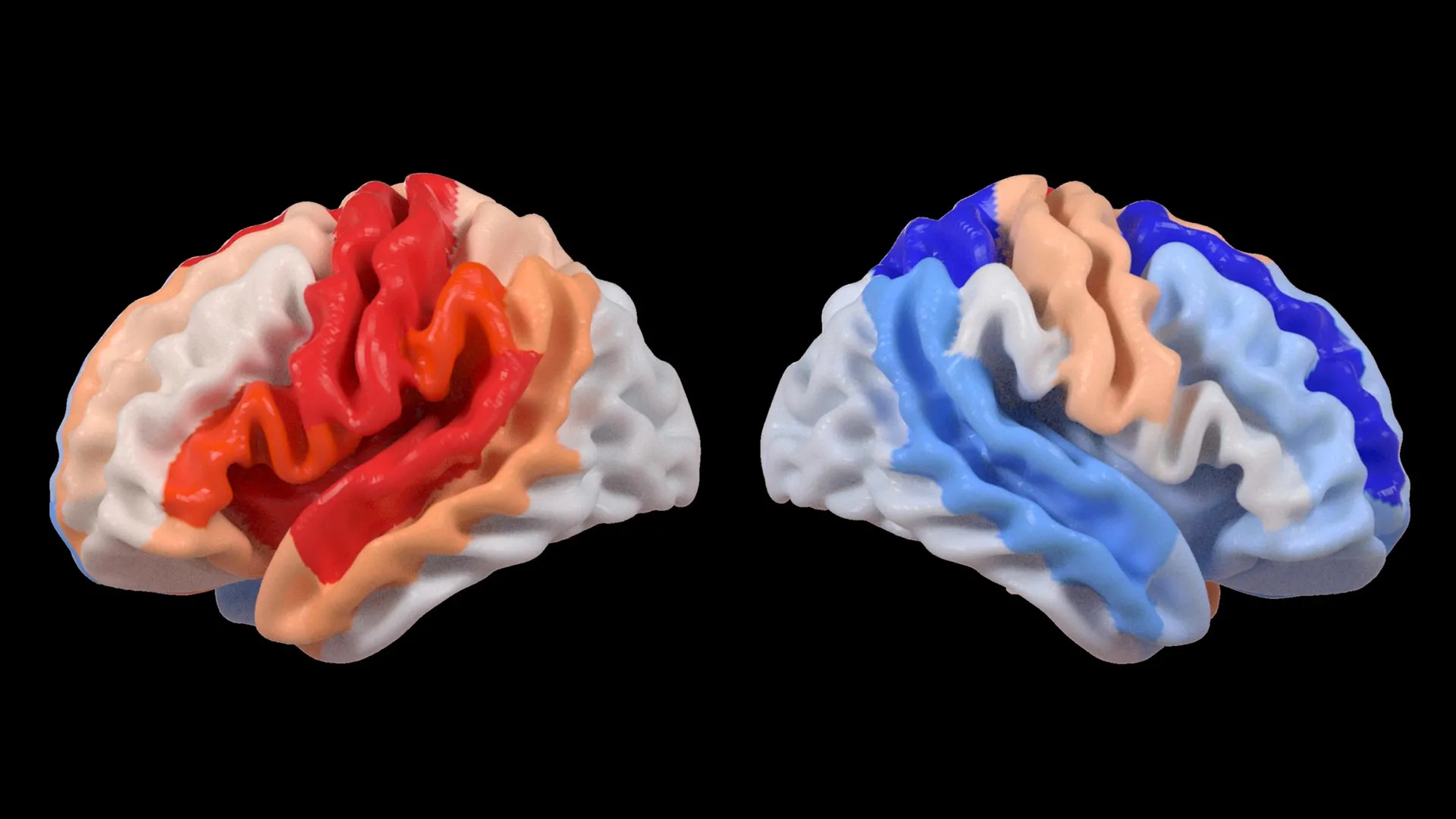
One of the most striking discoveries from this extensive analysis is the paradoxical observation concerning brain aging. Hosung Kim, PhD, an associate professor of research neurology at the Keck School of Medicine of USC and a co-senior author of the study, articulated this finding with remarkable clarity. "We found that larger strokes accelerate aging in the damaged hemisphere but paradoxically make the opposite side of the brain appear younger," Dr. Kim explained. "This pattern suggests the brain may be reorganizing itself, essentially rejuvenating undamaged networks to compensate for lost function."
This observation challenges conventional understanding of brain aging in the context of neurological injury. Typically, severe brain damage is associated with accelerated aging across affected areas. However, this study indicates that in stroke survivors, particularly those with significant motor deficits, the brain’s compensatory mechanisms might involve a rejuvenation process in the contralateral (opposite) hemisphere. This rejuvenation is not a literal reversal of biological age but rather a structural signature that the AI models interpret as indicative of a younger brain state.
Harnessing Artificial Intelligence for Neuroplasticity Insights
The sophisticated analysis underpinning these findings was made possible by a cutting-edge application of artificial intelligence. Specifically, the researchers utilized a type of AI known as a graph convolutional network. This powerful system was instrumental in estimating the biological age of 18 distinct brain regions based on the detailed MRI data. By comparing this AI-predicted age with each participant’s chronological age, the team calculated a metric called the brain-predicted age difference (brain-PAD). This difference serves as a valuable indicator of overall brain health, with a younger-than-expected brain age potentially signifying greater resilience or adaptive capacity.
The critical link between these brain age estimations and functional recovery emerged when the researchers correlated brain-PAD scores with motor function assessments. A significant pattern became apparent: stroke survivors who experienced severe impairments in movement, even after more than six months of dedicated rehabilitation, exhibited a younger-than-expected brain age in brain regions located on the side opposite to the initial stroke. This effect was particularly pronounced in the frontoparietal network, a critical brain system involved in a wide array of higher-order cognitive functions, including movement planning, attention, and complex coordination.
"These findings suggest that when stroke damage leads to greater movement loss, undamaged regions on the opposite side of the brain may adapt to help compensate," Dr. Kim elaborated. "We saw this in the contralesional frontoparietal network, which showed a more ‘youthful’ pattern and is known to support motor planning, attention, and coordination. Rather than indicating full recovery of movement, this pattern may reflect the brain’s attempt to adjust when the damaged motor system can no longer function normally. This gives us a new way to see neuroplasticity that traditional imaging could not capture."
The implications of this AI-driven insight are profound. It suggests that the brain, even in the chronic phase after a stroke, is not static but actively engaged in a process of self-reorganization. The observed "youthfulness" in the contralesional hemisphere may represent a rewiring effort, where these undamaged areas are recruited to take on or assist with functions that were previously handled by the injured hemisphere. This phenomenon highlights the brain’s remarkable plasticity, its ability to change and adapt in response to experience and injury.
The Power of Global Collaboration: The ENIGMA Initiative
The success of this study is intrinsically linked to the ENIGMA consortium, a global collaborative effort that exemplifies the power of big data in neuroscience. ENIGMA brings together researchers from over 50 countries, pooling their expertise and resources to foster a deeper understanding of the human brain across a spectrum of neurological conditions. By standardizing MRI data acquisition protocols and clinical information from numerous research groups, the ENIGMA initiative has successfully created one of the largest and most comprehensive stroke neuroimaging datasets ever assembled.
Arthur W. Toga, PhD, director of the Stevens INI and Provost Professor at USC, emphasized the transformative impact of such large-scale collaborations. "By pooling data from hundreds of stroke survivors worldwide and applying cutting-edge AI, we can detect subtle patterns of brain reorganization that would be invisible in smaller studies," Dr. Toga stated. "These findings of regionally differential brain aging in chronic stroke could eventually guide personalized rehabilitation strategies."
The sheer volume and diversity of the data analyzed in this study are crucial. Individual studies, while valuable, often suffer from limitations in sample size and generalizability. The ENIGMA approach, by aggregating data from a wide range of populations and clinical settings, mitigates these limitations. This allows for the identification of robust and consistent patterns that are more likely to reflect universal principles of brain adaptation after stroke. The ability to detect these subtle patterns of neuroplasticity, which might be overlooked by traditional imaging techniques, underscores the growing importance of advanced computational methods in modern neuroscience research.
Implications for Personalized Stroke Recovery Strategies
The findings from this study hold significant promise for the future of stroke rehabilitation and patient care. The ability to identify specific patterns of brain reorganization, particularly the apparent "rejuvenation" in undamaged areas, could pave the way for highly personalized treatment approaches. Currently, stroke rehabilitation often follows standardized protocols, which may not be optimal for every individual. By understanding how a patient’s brain is adapting, clinicians could potentially tailor therapies to leverage these compensatory mechanisms more effectively.
The researchers are keen to build upon these foundational discoveries. Their future plans involve longitudinal studies, where they will track patients over extended periods, from the acute phase immediately following a stroke through the long-term recovery trajectory. By monitoring how brain aging patterns and structural changes evolve over time, they aim to develop predictive models. These models could help anticipate a patient’s recovery potential and guide the selection of the most effective rehabilitation interventions.
The ultimate goal is to move towards a more precision-medicine approach in stroke recovery. This would involve using advanced neuroimaging and AI analysis to create a detailed profile of each patient’s brain, identifying specific areas of damage, compensatory activity, and potential for improvement. Armed with this information, healthcare professionals could then design customized rehabilitation programs that are optimized for each individual’s unique neurobiological profile, thereby enhancing the likelihood of functional recovery and improving their overall quality of life.
Broader Context and Future Directions
The study’s reliance on the ENIGMA consortium highlights a broader trend in scientific research: the increasing recognition that complex biological questions require collaborative, multi-institutional efforts and the integration of diverse datasets. The challenge of understanding the brain, with its billions of neurons and trillions of connections, is immense. Initiatives like ENIGMA provide the necessary scale and breadth to tackle these challenges effectively.
The use of deep learning to estimate brain age is a rapidly advancing field. While the concept of "brain age" is a proxy for biological health and has been linked to various neurological and psychiatric conditions, its application in understanding stroke recovery is a novel and powerful advancement. The ability of AI to discern subtle structural differences that correlate with functional outcomes offers a new lens through which to view the brain’s response to injury.
The implications of this research extend beyond stroke. Similar AI-driven approaches to estimating brain age and identifying compensatory neuroplasticity could potentially be applied to other neurological conditions, such as traumatic brain injury, Alzheimer’s disease, and Parkinson’s disease. By understanding how the brain adapts and reorganizes, researchers may uncover new therapeutic targets and develop more effective interventions for a wide range of neurological disorders.
The study, titled "Deep learning prediction of MRI-based regional brain age reveals contralesional neuroplasticity associated with severe motor impairment in chronic stroke: A worldwide ENIGMA study," received crucial funding from the National Institutes of Health (NIH) under grant R01 NS115845. This vital support enabled the large-scale data collection and analysis. Furthermore, the research benefited from the invaluable contributions of international collaborators from esteemed institutions, including the University of British Columbia, Monash University, Emory University, and the University of Oslo, underscoring the global nature of this significant scientific endeavor.
For those interested in a visual explanation of the associations between contralesional neuroplasticity and motor impairment, the Stevens INI has produced an informative video, accessible at https://www.youtube.com/watch?v=PYyy0-E0PKM. This resource offers a valuable supplementary perspective on the complex mechanisms at play in stroke recovery.